��Ŀ����
13��1915��ŵ��������ѧ������Henry Bragg��Lawrence Bragg���Ա���������X���߶Ծ���ṹ�ķ��������Ĺ��ף���һ����ѧ��ͨ��X����̽����NaCl��KCl��MgO��CaO����ṹ���ƣ��������־���ľ������������±���
| ���� | NaCl | KCl | CaO |
| ������/��kJ•mol-1�� | 786 | 715 | 3401 |
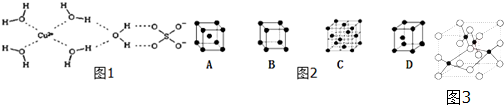
��������ѧ��ͨ��X�����Ʋ����������������������Ľṹʾ��ͼ1�ɼ�ʾ���£�
��1�����������к���ABC������ĸ���ţ�
A����λ�� B�����Ӽ� C�����Թ��ۼ�
D�������� E���� �� F���Ǽ��Թ��ۼ�
��2��Cu2+������NH3��Cl-���γ���λ��Ϊ4��������֪[Cu��NH3��4]2+���жԳƵĿռ乹�ͣ�[Cu��NH3��4]2+�е�����NH3������Cl-ȡ�����ܵõ����ֲ�ͬ�ṹ�IJ����[Cu��NH3��4]2+�Ŀռ乹��Ϊƽ�������Σ�
��3��ͼ2����ͭ��������C������ĸ���ţ��ѻ���ʽ��
��4��ͼ3��ͭ��ij��������ľ����ṹʾ��ͼ���þ�����ܶ�Ϊa g/cm3���谢��٤��������ֵΪNA����þ��������Ϊ$\frac{320}{aN{\;}_{A}}$cm3
���� ��һ��Ӱ�쾧�徧���ܴ�С�����������Ӱ뾶�Լ�����������ɵĶ��٣�������Խ���۵�Խ�ߣ�
��������1�����ݵ�������Ľṹͼ��֪���ھ�����ˮ������ͭ����֮������λ��������֮���Ǽ��Թ��ۼ����������ͭ����֮��Ϊ���Ӽ���ˮ������ˮ����֮���������
��2������[Cu��NH3��4]2+�е�����NH3������Cl-ȡ�����ܵõ����ֲ�ͬ�ṹ�IJ����жϣ�
��3��ͭ���������ܶѻ����ݴ��жϣ�
��4������V=$\frac{m}{��}$����������
��� �⣺��һ�����Ӱ뾶Mg2+��Na+��O2-��Ca2+��Cl-�����ӵ����Na+=Cl-��O2-=Mg2+=Ca2+�����Ӿ�������Ӱ뾶ԽС���������Խ�࣬������Խ��������۷е�Խ�ߣ�����NaCl��KCl��MgO��CaO�۵��ɸߵ��͵�˳����MgO��CaO��NaCl��KCl��
�ʴ�Ϊ��MgO��CaO��NaCl��KCl��
��������1�����ݵ�������Ľṹͼ��֪���ھ�����ˮ������ͭ����֮������λ��������֮���Ǽ��Թ��ۼ����������ͭ����֮��Ϊ���Ӽ���ˮ������ˮ����֮���������
��ѡABC��
��2������[Cu��NH3��4]2+�е�����NH3������Cl-ȡ�����ܵõ����ֲ�ͬ�ṹ�IJ��˵��Ϊƽ��ṹ��ƽ�������Σ��ʴ�Ϊ��ƽ�������Σ�
��3��ͭ���������ܶѻ������������ѻ���AΪ����������BΪ��������CΪ����������DΪ�����ѻ�����ѡC��
��4�����ݾ�̯����֪����ÿ�������к���ͭԭ����Ϊ8��$\frac{1}{8}$+6��$\frac{1}{2}$=4����ԭ����Ϊ4������V=$\frac{m}{��}$=$\frac{\frac{4����16+64��}{N{\;}_{A}}}{a}$cm3=$\frac{320}{aN{\;}_{A}}$cm3���ʴ�Ϊ��$\frac{320}{aN{\;}_{A}}$��
���� ���⿼������ݽ��ӣ��漰�����Ӿ���ľ����ܡ���������ṹ�������Ľṹ�������ļ����֪ʶ����һ�����ۺ��ԣ��е��Ѷȣ�

| A�� | ����ԭ�Ӳ�ͬ | B�� | ԭ�ӵĸ������з�ʽ��ͬ | ||
| C�� | ���в�ͬ�ļ������� | D�� | Ӳ�Ȳ�ͬ |
| A�� | CO32- | B�� | Ba2+ | C�� | Ag+ | D�� | Al3+ |
| A�� | ������Ԫ�ص����Ӳ�һ������������ | |
| B�� | ���������ӱ���ԭ��һ���õ��������� | |
| C�� | ��������ֻ�ܱ���ԭ�����ܱ����� | |
| D�� | ���������ﲻһ�����Ǽ��������� |
| A�� | Ư��¶���ڿ�����ʧЧ��ClO-+CO2+H2O�THClO+HCO3- | |
| B�� | ��NaAlO2��Һ��ͨ�����CO2��AlO2-+CO2+2H2O�TAl��OH��3��+HCO3- | |
| C�� | ̼�������Һ�����ᷴӦ��Ca��HCO3��2+2H+�TCa2++2H2O+2CO2�� | |
| D�� | ǿ����Һ��NaClO��Fe��OH��3��Ӧ����Na2FeO4��3ClO-+Fe��OH��3�TFeO42-+3Cl-+H2O+4H+ |
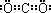 ��
�� ��������3��4һ��������
��������3��4һ�������� ��1mol������ȫȼ������������18.5mol��
��1mol������ȫȼ������������18.5mol��