��Ŀ����
һ���¶��£���2L���ܱ������м���4mol A��6mol B���������·�Ӧ��
2A��g��+3B��g�� 4C��g��+D��g����Ӧ10min���ƽ�⣬��ʱD��Ũ��Ϊ0.5mol/L������˵����ȷ����
4C��g��+D��g����Ӧ10min���ƽ�⣬��ʱD��Ũ��Ϊ0.5mol/L������˵����ȷ����
A��ǰ10min��Ӧ��ƽ������V��C��=0.10mol/L��min
B��10min���������м���A������ƽ��ʱ A��ת����һ������50%
A��ת����һ������50%
C�������½���Ӧ���������Сһ�룬��D��ƽ��Ũ��С��1.0mol/L
D����Ӧ��ƽ��ʱB��ƽ��Ũ����1.5mol/L
2A��g��+3B��g��
 4C��g��+D��g����Ӧ10min���ƽ�⣬��ʱD��Ũ��Ϊ0.5mol/L������˵����ȷ����
4C��g��+D��g����Ӧ10min���ƽ�⣬��ʱD��Ũ��Ϊ0.5mol/L������˵����ȷ���� A��ǰ10min��Ӧ��ƽ������V��C��=0.10mol/L��min
B��10min���������м���A������ƽ��ʱ
 A��ת����һ������50%
A��ת����һ������50%C�������½���Ӧ���������Сһ�룬��D��ƽ��Ũ��С��1.0mol/L
D����Ӧ��ƽ��ʱB��ƽ��Ũ����1.5mol/L
D
��ƽ��ʱD��Ũ��Ϊ0.5mol/L,����D��ʾ�ķ�Ӧ������V��D��=0.050mol/L��min����V��C��=0.20mol/L��min����A�����˷�ӦΪ���巴Ӧ����ѹƽ�ⲻ�ƶ�����10min���������м���A������ƽ��ʱ A��ת����һ������50%�������½���Ӧ���������Сһ�룬��D��ƽ��Ũ�ȵ���1.0mol/L������B��C����D�ԡ�
A��ת����һ������50%�������½���Ӧ���������Сһ�룬��D��ƽ��Ũ�ȵ���1.0mol/L������B��C����D�ԡ�
 A��ת����һ������50%�������½���Ӧ���������Сһ�룬��D��ƽ��Ũ�ȵ���1.0mol/L������B��C����D�ԡ�
A��ת����һ������50%�������½���Ӧ���������Сһ�룬��D��ƽ��Ũ�ȵ���1.0mol/L������B��C����D�ԡ�
��ϰ��ϵ�д�
�����Ŀ
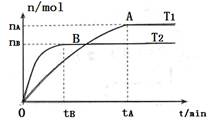
 CH3OH(g)+H2O(g)���������������������£��¶ȶԷ�Ӧ��Ӱ�죬��ͼ��ʾ��ע��T1��T2������300�棩������˵����ȷ����
CH3OH(g)+H2O(g)���������������������£��¶ȶԷ�Ӧ��Ӱ�죬��ͼ��ʾ��ע��T1��T2������300�棩������˵����ȷ����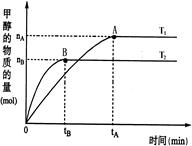
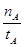 mol��L-1��min-1
mol��L-1��min-1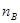 mol��L-1
mol��L-1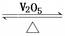 2SO3(g)����H��0��
2SO3(g)����H��0��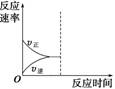
 CO2��g��+H2��g�� ��H��0�Իش��������⣺
CO2��g��+H2��g�� ��H��0�Իش��������⣺ 2/3Fe(s)+CO2(g)
2/3Fe(s)+CO2(g)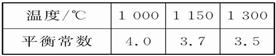
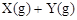
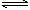
 ��
��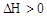 ������������ȷ���ǣ� ��
������������ȷ���ǣ� �� ����
����

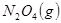 ����H
����H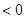 �����ܱ������н��У����ﵽƽ��ʱ����ͨ���ı��������ﵽ��ƽ���ʹ������ɫ���Ӧ��ȡ�Ĵ�ʩ��
�����ܱ������н��У����ﵽƽ��ʱ����ͨ���ı��������ﵽ��ƽ���ʹ������ɫ���Ӧ��ȡ�Ĵ�ʩ��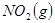
 CH3OH(g) ��H<0�������ʵ�Ũ�����±�
CH3OH(g) ��H<0�������ʵ�Ũ�����±�