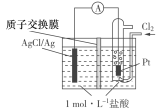
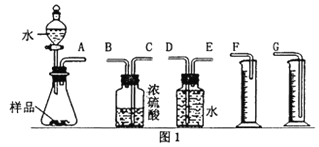
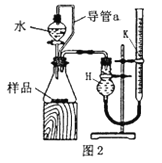
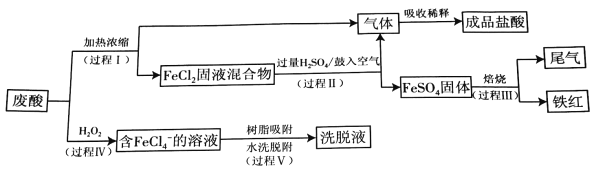
��Ŀ����
����Ŀ����Ϊ�ⶨij�л�������A�Ľṹ����������ʵ�飺
��1�����л���A����O2���г��ȼ�գ�ʵ���ã�����5��4 g H2O��8��8 g CO2������O26��72 L(��״����)����������и�Ԫ�ص�ԭ�Ӹ�������N(C)��N(H)��N(O)= ��
��2���������Dzⶨ���л����������Է��������������ʵķ���ʽ�� ��
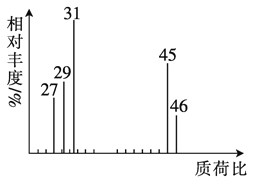
��3���˴Ź��������ܶ��л�������в�ͬ��ѧ��������ԭ�Ӹ�����ͬ�ķ�ֵ(�ź�)�����ݷ�ֵ(�ź�)����ȷ����������ԭ�ӵ��������Ŀ�����磺���ȼ���(ClCH2OCH3)��������ԭ����ͼ�������ⶨ���л���A�ĺ˴Ź�������ʾ��ͼ��ͼ������A�Ľṹ��ʽΪ ��

��4��A��һ����������ˮ������B��B�ɺϳɰ�װ����C����д��Bת��ΪC�Ļ�ѧ����ʽ�� ��
���𰸡���1��2��6��1��
��2��C2H6O��
��3��CH3CH2OH��
��4��![]() ��
��
�������������������1��5.4gH2OΪ![]() =0.3mol��
=0.3mol��
��n(H)=0.6mol��8.8gCO2Ϊ![]() =0.2mol��
=0.2mol��
��n(C)=0.2mol��6.72LO2Ϊ![]() =0.3mol��
=0.3mol��
��OԪ���غ��֪�л����к���n(O)=0.3mol+0.2mol��2-0.3mol��2=0.1mol��
��n(C)��n(H)��n(O)=2��6��1���ʴ�Ϊ��n(C)��n(H)��n(O)=2��6��1��
��2����n(C)��n(H)��n(O)=2��6��1�������л��������ʵ��ʽΪC2H6O�����ʺɱȿ�֪��Է�������Ϊ46�������ʽΪC2H6O���ʴ�Ϊ��C2H6O��
��3��A�Ŀ��ܽṹΪ��CH3CH2OH��CH3OCH3�����ⶨ���л���A�ĺ˴Ź�������ʾ��ͼ��ͼ����˵����3�����ʲ�ͬ��Hԭ�ӣ���AӦΪCH3CH2OH���ʴ�Ϊ��CH3CH2OH��
��4��A��һ����������ˮ������B��BӦΪCH2=CH2��B�ɺϳɰ�װ����C��CӦΪ����ϩ��Bת��ΪC�Ļ�ѧ����ʽΪ![]() ���ʴ�Ϊ��
���ʴ�Ϊ��![]() ��
��

����Ŀ��X��Y��Z��WΪ���ֶ�����Ԫ�أ�����YԪ��ԭ�Ӻ�������������������Ӳ�����3�������������ڱ��е����λ������ͼ��ʾ��
X | Y | |
Z | W |
��ش��������⣺
(1)Wλ�����ڱ��е�________���ڣ���______�壻
(2)X���γ�˫ԭ�ӷ��ӣ�����ӵĵ���ʽ��_________��Y����Ԫ���γɵ�10�������г���+1��������Ϊ_______���ѧʽ����ͬ����Z����Ԫ���γɵ�18�������г�����-1�������ӵ�ˮ�ⷽ��ʽ__________��
(3)��ҵ�Ͻ������W����ͨ�����ڵ�Z�����п��Ƶû�����Z2W2�������ʿ���ˮ��Ӧ����һ����ʹƷ����Һ��ɫ�����壬0.2mol�����ʲμӷ�Ӧʱת��0.3mol���ӣ�����ֻ��һ��Ԫ�ػ��ϼ۷����ı䣬д��Z2W2��ˮ��Ӧ�Ļ�ѧ����ʽ_________________________��
(4)��0.20molYZ2��0.10molO2����һ���̶��ݻ�Ϊ5L���ܱ������У���һ���¶Ȳ��д��������£����з�Ӧ��������Ӵﵽƽ�⣬�����Һ�к�YZ30.18mol����v(O2)=________mol/(L��min)�����¶Ȳ��䣬����ͨ��0.20molYZ2��0.10molO2����ƽ��_______�ƶ����������Ӧ���������淴Ӧ�����������ٴδﵽƽ���________mol<n(YZ3)<_______mol��