��Ŀ����
��ѧ��Ӧ�м������ʱ仯�����������仯���ͷŻ����������ǻ�ѧ��Ӧ�������仯����Ҫ��ʽ֮һ����֪C(ʯī)��H2(g)ȼ�յ��Ȼ�ѧ����ʽ�ֱ�Ϊ��
�� C(ʯī)+ O2(g)��CO(g)
O2(g)��CO(g) 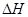 ="-111.0" KJ��mol-1
="-111.0" KJ��mol-1
�� H2(g)+  O2(g) ��H20(g)
O2(g) ��H20(g) 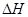 ="-242.0" kJ��mol-1
="-242.0" kJ��mol-1
�� C(ʯī)+O2(g)��CO2(g) 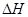 ="-394.0" kJ��mol-1
="-394.0" kJ��mol-1
�����������⣺
��1����ѧ��Ӧ���������仯�ı���ԭ���Ƿ�Ӧ�������� �Ķ��Ѻ��γɡ�����������Ӧ���� (����ȡ����ȡ�)��Ӧ��
��2�����Ȼ�ѧ����ʽ�У���Ҫ������Ӧ�P�������״̬��ԭ���� ���ڢ��У�02�Ļ�ѧ��������1/2���DZ�ʾ (����ĸ)��
a�����Ӹ��� b�����ʵ��� c����������
��3����Ӧ2H20(g)��2H2(g)+02(g)��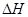 = KJ��mol-1��
= KJ��mol-1��
��4����C(���ʯ)+02(g)��C02(g)��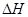 ="-395.0" kJ��mol-1�����ȶ��ԣ����ʯ (�����������������)ʯī��
="-395.0" kJ��mol-1�����ȶ��ԣ����ʯ (�����������������)ʯī��
��5����֪�γ�H20(g)�е�2 mol H-O���ܷų�926.0 kJ���������γ�1 mol 02(g)�еĹ��ۼ��ܷų�498.0 kJ�������������1 mol H2(g)�е�H-H����Ҫ������ KJ��
��6����ҵ��������һ����Ҫ;������CO(g)��H2O(g)��Ӧ����C02(g)��H2(g)����÷�Ӧ���Ȼ�ѧ����ʽ�� ��
��1����ѧ�� ����
��2����Ӧ���뷴Ӧ�P�������״̬�������йأ������𰸾��ɣ� b
��3��484.0 ��4���� ��5��435.0
��6��CO(g)��H2O(g)��CO2(g)��H2(g) ��H����41 kJ��mol��1
���������������2������״̬��ͬ�����е�������ͬ��״̬֮���ת�������������ı仯�������Ȼ�ѧ����ʽ�б���������ʵ�״̬����3���÷�Ӧ��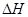 �Ƿ�Ӧ�ڵ�2�����෴�����ɷ���ʽ�жϣ����Ȼ�ѧ����ʽ�еļ�������ָ���ʵ�����û���۵ĺ��壻��4�����ʺ��е�����Խ�࣬�ȶ���Խ����ʯ����ʯī�ų��������ó����ʯ���������ߣ��ȶ��Բ
�Ƿ�Ӧ�ڵ�2�����෴�����ɷ���ʽ�жϣ����Ȼ�ѧ����ʽ�еļ�������ָ���ʵ�����û���۵ĺ��壻��4�����ʺ��е�����Խ�࣬�ȶ���Խ����ʯ����ʯī�ų��������ó����ʯ���������ߣ��ȶ��Բ
��5������1 mol H2(g)�е�H-H����Ҫ������=926.0-498.0��2- 242.0 ="435.0" KJ��
��6����Ӧ�ۣ��٣��ڵõ�CO(g)��H2O(g)��CO2(g)��H2(g) ��H����41 kJ��mol��1
���㣺���鷴Ӧ���й����⡣

�״���һ�ֿ�������Դ�����й㷺�Ŀ�����Ӧ��ǰ����
(1)��ҵ��һ������������ַ�Ӧ�ϳɼ״���
��Ӧ��CO(g)��2H2(g) CH3OH(g)����H1
CH3OH(g)����H1
��Ӧ��CO2(g)��3H2(g) CH3OH(g)��H2O(g)����H2
CH3OH(g)��H2O(g)����H2
��������Ӧ���ϡ�ԭ�Ӿ��á�ԭ�����________(���)��
���±����������Ƿ�Ӧ���ڲ�ͬ�¶��µĻ�ѧƽ�ⳣ��(K)��
| �¶� | 250 �� | 300 �� | 350 �� |
| K | 2.041 | 0.270 | 0.012 |
�ɱ��������жϣ���H1______0(�����������������)��
��ij�¶��£���2 mol CO��6 mol H2����2 L���ܱ������У���ַ�Ӧ���ﵽƽ����c(CO)��0.2 mol��L��1����CO��ת����Ϊ________����ʱ���¶�Ϊ________(���ϱ���ѡ��)��
(2)��֪�ڳ��³�ѹ�£�
��2CH3OH(l)��3O2(g)===2CO2(g)��4H2O(g) ��H1����1 275.6 kJ��mol��1
��2CO(g)��O2(g)===2CO2(g) ��H2����566.0 kJ��mol��1
��H2O(g)===H2O(l)����H3����44.0 kJ��mol��1
д���״�����ȫȼ������һ����̼��Һ̬ˮ���Ȼ�ѧ����ʽ��__________��
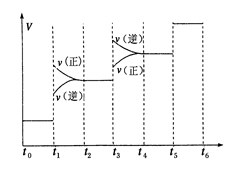
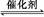 2CO2��g��+N2��g�������ܱ������з����÷�Ӧʱ,c��CO2�����¶ȣ�T���������ı������S����ʱ�䣨t���ı仯����,��ͼ��ʾ��
2CO2��g��+N2��g�������ܱ������з����÷�Ӧʱ,c��CO2�����¶ȣ�T���������ı������S����ʱ�䣨t���ı仯����,��ͼ��ʾ��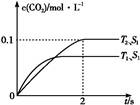

 N2��g��+CO2��g��+2H2O��g����H="-867" kJ/mol
N2��g��+CO2��g��+2H2O��g����H="-867" kJ/mol N2O4��g������H="-56.9" kJ/mol
N2O4��g������H="-56.9" kJ/mol
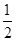 O2(g)=H2O(g) ��H1����241.8 kJ��mol��1
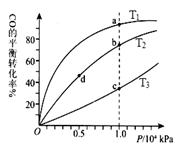
O2(g)=H2O(g) ��H1����241.8 kJ��mol��1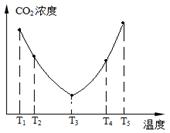
 CH3OH ( g ) ��H��-90.8 kJ��mol��1 ��һ�ݻ��ɱ���ܱ������г���10 mol CO ��20 molH2��CO ��ƽ��ת�������¶ȣ�T����ѹǿ��P���ı仯��ͼ��ʾ�����ﵽƽ��״̬A ʱ�����������Ϊ20 L��
CH3OH ( g ) ��H��-90.8 kJ��mol��1 ��һ�ݻ��ɱ���ܱ������г���10 mol CO ��20 molH2��CO ��ƽ��ת�������¶ȣ�T����ѹǿ��P���ı仯��ͼ��ʾ�����ﵽƽ��״̬A ʱ�����������Ϊ20 L��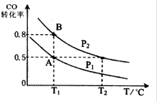
 CO��g��+H2O��g�� ��H=" +" 41.3 kJ��mol��1�������
CO��g��+H2O��g�� ��H=" +" 41.3 kJ��mol��1������� SO3��g����NO��g��
SO3��g����NO��g��
 2NH3(g)����H����92.4 kJ/mol
2NH3(g)����H����92.4 kJ/mol