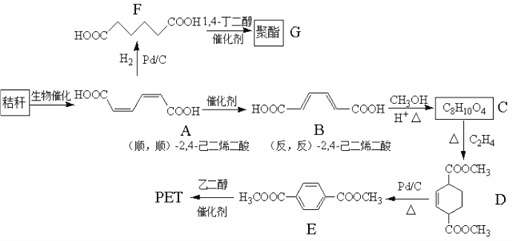
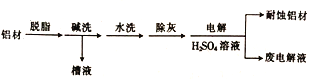
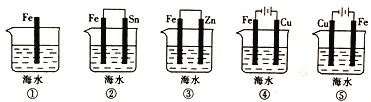
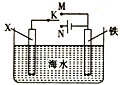
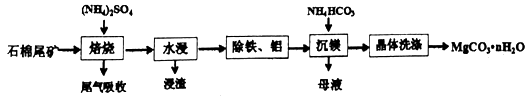
��Ŀ����
����Ŀ��һ���¶��£���һ��10 L�ܱ������з���ij���淴Ӧ����ƽ�ⳣ������ʽΪK��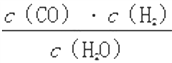 ����ش��������⡣
����ش��������⡣
��1�����¶����ߣ�K������÷�Ӧ��________��Ӧ(��������������������)��
��2�����жϸ÷�Ӧһ���ﵽƽ��״̬����________(����ĸ���)��
A��v��(H2O)��v��(H2)
B�������������ƽ����Է�����������ʱ��ı�
C������n mol H2��ͬʱ����n mol CO
D�����������ʵ������ʵ�������ʱ��ı�
���𰸡� ���� AB
��������(1)�¶����ߣ�K����˵��ƽ�������ƶ����������ӦΪ���ȷ�Ӧ���ʴ�Ϊ�����ȣ�
(2)����ƽ�ⳣ���Ķ��壬������Ũ�ȵ���֮���뷴Ӧ��Ũ�ȵ���֮��֮��Ϊƽ�ⳣ������Ӧ�ķ���ʽΪC(s)+H2O(g)CO(g)+H2 (g)��A������(H2O)=����(H2)ʱ�����淴Ӧ������ͬ����Ӧ�ﵽƽ�⣬��A��ȷ��B����Ӧǰ����������ʵ�������ͬ�����������仯�������������ƽ����Է�����������ʱ��仯���ﵽƽ�⣬��B��ȷ��C������nmolH2��ͬʱ����nmolCO����Ӧ�������淴Ӧ������˵����Ӧ�ﵽƽ��״̬����C����D�����������ʵ������ʵ���ʼ�ղ��䣬��D���ʴ�Ϊ��AB��

����Ŀ����1�������£���0.1000mol/LCH3COOH��Һ�ζ�NaOH��Һ���ﵽ�ζ��յ�ʱ����Һ�и�����Ũ�ȴӴ�С��˳��Ϊ________��Ϊ�˼�С�ζ�����ڵζ�ʵ����ʹ�õ����ָʾ�������________������ţ�
A��ʯ�� B������ C����̪ D�����Ȼ��̪
��2�������£���pH��5��H2SO4��Һ����ˮ�������H��Ũ��ΪC1��pH��5��Al2(SO4)3��Һ����ˮ�������H��Ũ��ΪC2����C1/C2��________��
��3����֪H2A�ĵ��뷽��ʽΪ��H2A=H++HA-��HA-![]() H++A2-�����ж�H2A��_________������ǿ��������������ʡ�
H++A2-�����ж�H2A��_________������ǿ��������������ʡ�
��4����֪CH3COOH���ܼ�A�п���ȫ�����룬�β��ܽ���A�ܼ�����CH3COOH��Na2CO3���ܼ�A������CO2��Ӧ�����ӷ���ʽ��________��
��5������ƽ�ⳣ���Ǻ���������ʵ���̶ȵ�����������֪��
��ѧʽ | ���볣��(25 ��) |
HCN | K��4.9��10��10 |
CH3COOH | K��1.8��10��5 |
H2CO3 | K1��4.3��10��7��K2��5.6��10��11 |
��25 ��ʱ���е�Ũ�ȵ�HCN��Һ��H2CO3��Һ��CH3COOH��Һ������Һ��pH�ɴ�С��˳��Ϊ__________________(�û�ѧʽ��ʾ)��
����NaCN��Һ��ͨ��������CO2��������Ӧ�Ļ�ѧ����ʽΪ__________________��