��Ŀ����
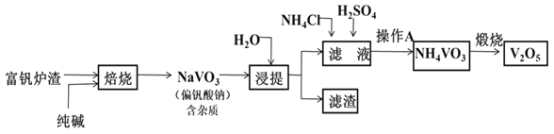
����Ŀ���о����֣�����Խϡ����ԭ�����е�Ԫ�صĻ��ϼ�Խ�͡�ijͬѧȡ���������ۺ����۵Ļ������������ϡ�������ַ�Ӧ��������ų����ڷ�Ӧ���������Һ�У���μ���4mol/LNaOH��Һ������NaOH��Һ�����(ml)������ij��������ʵ���(mol)�Ĺ�ϵ��ͼ��ʾ������˵������ȷ���ǣ� ��

A.��Һ��n(NH4+)=0.012mol
B.��������������������ʵ����ı�Ϊ1��2
C.�ⶨF������������������ǣ����ˡ�ϴ�ӡ����º�ɡ�����
D.�����Ũ�����ᷴӦ�����ӷ���ʽΪ��8Al+30H++3NO3-=8Al3++3NH4++9H2O
���𰸡�B
��������
���ۺ����۵Ļ������������ϡHNO3��ַ�Ӧ��������ΪAl3+��Fe3+��ͨ�����⣬��Ӧʼ��û���������ɣ����Եó������е������������ɣ����������Ũ��Խϡ����Ӧ��ԭ�����е�Ԫ�صĻ��ϼ�Խ�ͣ������Ʋ�NԪ����+5�����-3�ۣ���ͼ�ɵ������������������������ҺӦ�������ᷴӦ�������ɳ�������������ȫ����ͼ֪������������������Һ�����������䣬�ɵ���NH4+�����˷�Ӧ��������NaOH�ĵμӣ������ķ�Ӧ�����У���H++OH-=H2O����Fe3++3OH-=Fe(OH)3����Al3++3OH-=Al(OH)3������NH4++OH-�TNH3H2O����Al(OH)3+OH-=AlO2-+2H2O��
A�� ��ͼ��֪DE�������������Ƶ����Ϊ3ml����NH4++OH�TNH3H2O��֪��Һ��n(NH4+)=n(OH)=0.003L��4mol/L=0.012mol����A��ȷ��
B����ͼ��֪��DE�����ĵ��������Ƶ����Ϊ34mL31m=3mL���ʸýβμӷ�Ӧ����������Ϊ0.003L��4mol/L=0.012mol������NH4++OH�TNH3H2O��֪��������Һ��n(NH4+)=0.012ml������EF�η���Al(OH)3+OH=AlO2+2H2O����֪n[Al(OH)3]=0.008mol��������Ԫ���غ㣬�ʻ�Ͻ�����n(Al)=0.008mol�����ݵ���ת���غ���3n(Fe)+3n(Al)=8n(NH4+)����3n(Fe)+3��0.008mol=8��0.012mol�����n(Fe)=0.024mol����ԭ����������������۵����ʵ���֮��Ϊ0.008mol��0.024mol=1:3����B����
C�����ⶨF�������������Ӧ���������ܵĹ�����Һ����룬��ϴ�ӳ������ŵ��ӻ����ٸ����������ʵ�鲽���ǣ����ˡ�ϴ�ӡ������������C��ȷ��
D��Al��ϡ���ᷴӦ����������������泥����ӷ�Ӧ����ʽ��8Al+30H++3NO3-=8Al3++3NH4++9H2O����D��ȷ��
��ѡB��

 ��ѧ��ʦ����ϵ�д�
��ѧ��ʦ����ϵ�д�