��Ŀ����
13�����Ȼ�����S2Cl2���ڹ�ҵ����������Ϊ��ʵ���Һϳ�S2Cl2��ij��ѧ�о���ѧϰС��������й����ϣ��õ�������Ϣ��| ���� | �۵�/�� | �е� | ��ѧ���� |
| S | 112.8 | 444.6 | �� |
| S2Cl2 | -77 | 137 | ��ˮ����HCl��SO2��S��300��������ȫ�ֽ⣬S2Cl2+Cl$\frac{\underline{\;\;��\;\;}}{\;}$ 2SCl2 |
���й����ʵIJ����������ϱ���
���ʵ��װ��ͼ���£�
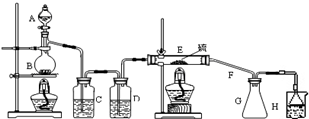
��ش��������⣺
��1��B �з�Ӧ�����ӷ���ʽMnO2+4H++2Cl- $\frac{\underline{\;\;��\;\;}}{\;}$Mn2++Cl2��+2H2O��E �з�Ӧ�Ļ�ѧ����ʽ2S+C12$\frac{\underline{\;\;��\;\;}}{\;}$S2C12��
��2��C��D �е��Լ��ֱ��DZ���ʳ��ˮ��Ũ���ᣮ
��3������ڼ���Eʱ�¶ȹ��ߣ���ʵ������Ӱ����S2Cl2���¶ȹ���ֽ⣮
��4������S2Cl2�����ʣ�����ʵ��װ����G��H֮��Ӧ���Ӹ���װ�ã�
���� ��1��B�з�����Ӧ�����ӷ���ʽ��Ũ���������������ȡ���������ӷ���ʽ��E�з�Ӧ�Ļ�ѧ����ʽ����Ϣ�������������110�桫140������Ӧ�����ɵ�S2Cl2 ��Ʒ��
��2��������ȡ�����еij������⣬���ñ���ʳ��ˮ��ȥ�����е�HCl������Ũ������и��T�ɵô��������������
��3���ɱ�����Ϣ S2Cl2��300��������ȫ�ֽ��ǣ�
��4������Ϣ��֪S2Cl2��ˮˮ�⣬H������������Һ��ˮ�����ӷ�����G�У�
��� �⣺��1��B�з�����Ӧ�����ӷ���ʽ��Ũ���������������ȡ���������ӷ���ʽ��MnO2+4H++2Cl- $\frac{\underline{\;\;��\;\;}}{\;}$Mn2++Cl2��+2H2O�������������110�桫140������Ӧ�����ɵ�S2Cl2 ��Ʒ������ʽΪ��2S+C12$\frac{\underline{\;\;��\;\;}}{\;}$S2C12��
�ʴ�Ϊ��MnO2+4H++2Cl-$\frac{\underline{\;\;��\;\;}}{\;}$Mn2++Cl2��+2H2O��2S+C12$\frac{\underline{\;\;��\;\;}}{\;}$S2C12��
��2��������ȡ�����еIJ�������HCl��H2O���������Cװ�ã�����ʳ��ˮ����ȥ�����е�HCl������Dװ�ã�Ũ���ᣩ���и��T�ɵô��������������
�ʴ�Ϊ������ʳ��ˮ��Ũ���
��3���ɱ�����Ϣ S2Cl2��300��������ȫ�ֽ⣬���²��ʽ��ͣ�
�ʴ�Ϊ��S2Cl2���¶ȹ���ֽ⣻
��4��G�ռ���Ʒ��H������������Һ��ˮ�����ӷ�������G�У���ʹS2Cl2ˮ�⣬��G��H֮�����Ӹ���װ�ã�
�ʴ�Ϊ������װ�ã�
���� ������Ҫ��������ʵ�����кϳ�S2C12��������Ŀ��Ϣ����ԭ��������ѧ������֪ʶ����������������������ۺ��Խ�ǿ���Ѷȴ�

| A�� | ��������NaOH��Һ��Ӧ���Ҵ����� | |
| B�� | ��Na��Ӧʱ���Ҵ��ķ�Ӧ���ʱ�ˮ�� | |
| C�� | �Ҵ��ܱ������ظ������Һ����������� | |
| D�� | �����ױ�����������Ӧ����һ����ȡ������ʱ���ױ��ķ�Ӧ�¶ȸ��� |

��֪����Cu2O+2H+=Cu+Cu2++H2O
�������������������pH��
| ������ | Cu��OH��2 | Al��OH��3 | Fe��OH��3 | Fe��OH��2 |
| ��ʼ����pH | 5.4 | 4.0 | 1.1 | 5.8 |
| ������ȫpH | 6.7 | 5.2 | 3.2 | 8.8 |
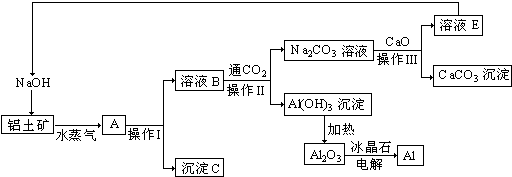
��2����֤ú�ۻҺ���FeO��Ҫ�Ļ�ѧ�Լ�Ϊϡ���ᡢKMnO4��Һ ����ϡ���ᡢK3[Fe��CN��6]��Һ����
��3�����������Ҫ�ɷ�ΪCu��SiO2���ѧʽ�������������Ҫ�ɷ�ΪFe ��OH��3��Cu��OH��2���ѧʽ��
��4������X�Ļ�ѧʽΪCO2�������ܡ�ʱ��Ӧ�����ӷ���ʽΪAl��OH��3+OH-=AlO2-+2H2O��
��5��Ϊ�˻�ò�ƷAl��OH��3���ӷ�ú�ҵ������ȡ�����Һ��ʼ����ֻ��CaCO3һ���Լ����������������Ǽ���CaCO3����pH��3.2�����˳�ȥFe��OH��3���ټ���CaCO3����pH��5.2�����˵õ�Al��OH��3_��
| A | C | |
| B | D |
| A�� | ��̬�⻯����ȶ��ԣ�HC��HD | |
| B�� | ����������Ӧˮ��������ԣ�H2BO4��HDO4 | |
| C�� | ���ʷе���ߵ���BԪ���γɵĵ��� | |
| D�� | A����̬�⻯��ˮ��Һ�Լ��� |
| A�� | -CHO �ʻ� | B�� | -OH �ǻ� | C�� | -COOH ȩ�� | D�� | -NH2 ���� |
| A�� | �ܽ⡢���ˡ�������ϴ�� | B�� | �ܽ⡢������ϴ�ӡ����� | ||
| C�� | �ܽ⡢���ˡ�ϴ�ӡ����� | D�� | �ܽ⡢ϴ�ӡ����ˡ����� |