��Ŀ����
14��ij�л���A����Է�������Ϊ128���ʣ���1����A��ֻ����C��HԪ�أ���A���ӽṹ�к��б�������A�Ļ�ѧʽΪC10H8��
��2����A��ֻ����C��H��OԪ�أ���A�����ﺬһ�����������������������ţ���A�Ļ�ѧʽΪC7H12O2��д������һ�ַ�����������ʵĽṹCH3COOCH2CH2CH2CH=CH2��
��3����A��ֻ����C��H��NԪ�أ���A�����е�ԭ�������٣���A�Ļ�ѧʽΪC7H16N2��
���� ��1�������ķ���ʽΪCxHy��������̼ԭ�������Ŀ=$\frac{128}{12}$=10��8������ʽΪC10H8������1��Cԭ����Ҫ����12��Hԭ�ӣ�����ʽ��ΪC9H20��C9H20�DZ��������������������Hԭ����Ŀ��
��2��A�����ﺬһ������-COO-����Է�������Ϊ44�����������������ţ���ʣ��IJ���Ϊ����������������Է�������Ϊ128-44=84�����е�̼ԭ����Ŀ���=$\frac{84}{12}$=7�������ڲ�����ȫ����Cԭ�ӣ�������1��Cԭ����Ҫ����12��Hԭ�ӣ��������ķ���ʽ��ΪC6H12���ݴ˷���������Ļ�ѧʽ�Ϳ��ܵĽṹ��
��3����A�к���C��H��NԪ�أ���Nԭ�ӻ���-NH2����ʽ���ڣ���������Ϊ����-NH2ȡ����������ϩ����Ȳ���е���ԭ�Ӷ��γɣ��ݴ˷�����
��� �⣺��1�������ķ���ʽΪCxHy��������̼ԭ�������Ŀ=$\frac{128}{12}$=10��8������ʽΪC10H8������1��Cԭ����Ҫ����12��Hԭ�ӣ�����ʽ��ΪC9H20��C9H20�DZ��������������������Hԭ����Ŀ����A���ӽṹ�к��б�������A�ķ���ʽΪC10H8���ʴ�Ϊ��C10H8��
��2��A�����ﺬһ������-COO-����Է�������Ϊ44�����������������ţ���ʣ��IJ���Ϊ����������������Է�������Ϊ128-44=84�����е�̼ԭ����Ŀ���=$\frac{84}{12}$=7�������ڲ�����ȫ����Cԭ�ӣ�������1��Cԭ����Ҫ����12��Hԭ�ӣ��������ķ���ʽ��ΪC6H12�����������п��ܺ�����һ��̼̼˫�����Ӷ��ó����л���ķ���ʽΪC7H12O2�����ܵĽṹΪ��CH3COOCH2CH2CH2CH=CH2���ʴ�Ϊ��C7H12O2��CH3COOCH2CH2CH2CH=CH2��
��3����A�к���C��H��NԪ�أ���Nԭ�ӻ���-NH2����ʽ���ڣ���������Ϊ����-NH2ȡ����������ϩ����Ȳ���е���ԭ�Ӷ��γɣ�
���������к�һ��-NH2���������ʿ�����Ϊ����1��-NH2ȡ����������ϩ����Ȳ���е�1����ԭ�Ӷ��γɣ���ʣ�������Ӧ����-CnH2n+1��-CnH2n-1��CnH2n-3������������Է�������Ϊ128-16=112�����е�̼ԭ�ӵĸ������Ϊ$\frac{112}{12}$=9��4��������1��̼ԭ�ӣ�����ԭ�Ӽ�12����������Σ���ԭ�Ӿ�Ϊż������������ͨʽ-CnH2n+1��-CnH2n-1��CnH2n-3�����������в���ֻ��һ��-NH2��
���������к�2��-NH2���������ʿ�����Ϊ����2��-NH2ȡ����������ϩ����Ȳ���е�2����ԭ�Ӷ��γɣ���ʣ�������Ӧ����-CnH2n��-CnH2n-2��CnH2n-4������������Է�������Ϊ128-32=96�����е�̼ԭ�ӵĸ������Ϊ$\frac{96}{12}$=8����������ֻ��Cԭ����Hԭ�ӣ��ʼ���1��̼ԭ�ӣ�����ԭ�Ӽ�12��������Ϊ-C7H12������-CnH2n-2���ʴ��л���ΪC7H12��NH2��2��������ʽΪ��C7H16N2���ʴ�Ϊ��C7H16N2��
���� ���⿼���л�����ƶϵȣ�ע���������෨����෨ȷ���л���Ľṹ���Ѷ��еȣ�

 ��˼ά������ҵ���ټ��ִ�ѧ������ϵ�д�
��˼ά������ҵ���ټ��ִ�ѧ������ϵ�д�| A�� | ��״���£�2.24 L HF���е�HF������Ϊ0.1NA | |
| B�� | �ڳ��³�ѹ�£�32 g 18O2�к���2NA��ԭ�� | |
| C�� | 25��ʱ��pH=13��1.0 L Ba��OH��2��Һ�к��е�OH-��ĿΪ0.2NA | |
| D�� | 1 mol Na������O2��Ӧ������Na2O��Na2O2�Ļ���ת�Ƶĵ�����ΪNA |
| A�� | ԭ�Ӱ뾶�ɴ�С��˳��W��Z��Y��X�������Ӱ뾶�ɴ�С��˳��X��Y��Z | |
| B�� | ԭ�������������ɶൽ�ٵ�˳��Y��X��W��Z����������ɸߵ���˳��W��Z | |
| C�� | Ԫ�طǽ�������ǿ������˳��Z��W��X��W�ĵ��ʳ����¿�������������Һ��Ӧ | |
| D�� | ����̬�⻯����ȶ�����ǿ������˳��X��Y��W |
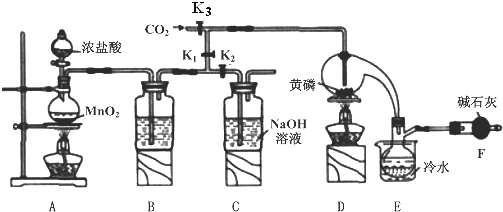
��֪��
�ٻ���������Cl2��Ӧ����PCl3�������Cl2��Ӧ����PCl5��
��PCl3��ˮ��ǿ��ˮ������H3PO3��HC1��
��PCl3��O2������POCl3��POCl3����PCl3��
��PCl3��POCl3���۷е������
| ���� | �۵�/�� | �е�/�� |
| PCl3 | -112 | 75.5 |
| POCl3 | 2 | 105.3 |
��1��Aװ���������������ӷ���ʽΪMnO2+4H++2Cl-$\frac{\underline{\;\;��\;\;}}{\;}$Mn2++Cl2��+2H2O��
��2��B����װ�Լ���Ũ���ᣬF�м�ʯ�ҵ����������֣��ֱ������ն������������ֹ�����е�H2O������ƿ��PCl3 ��Ӧ��
��3��ʵ��ʱ�����װ�������Ժ��ȴ�K3ͨ������CO2����Ѹ�ټ�����ף�ͨ����CO2���������ž�װ���еĿ�������ֹ������ȼ��
��4���ֲ�Ʒ�г�����POCl3��PCl5�ȣ���������ȳ�ȥPCl5��ͨ��������ʵ��������ƣ������ɵõ��ϴ�����PCl3��
��5��ʵ�����ʱ����������C�е��Լ����ն����������C�з�Ӧ�����ӷ���ʽΪCl2+2OH-=Cl-+ClO-+2H2O��
��6��ͨ�����淽���ɲⶨ��Ʒ��PCl3����������
��Ѹ�ٳ�ȡ1.00g��Ʒ����ˮ��Ӧ�����250mL��Һ��
��ȡ������Һ25.00mL�������м���10.00mL 0.1000mol/L��ˮ����ַ�Ӧ��
�����������Һ�м��뼸�ε�����Һ����0.1000mol/L��Na2S2O3��Һ�ζ���
���ظ��ڡ��۲���ƽ������Na2S2O3��Һ8.40mL��
��֪��H3PO3+H2O+I2�TH3PO4+2HI��I2+2Na2S2O3�T2NaI+Na2S4O6������ⶨ������û��������Ӧ�������������ݣ��ò�Ʒ��PC13����������Ϊ79.75%��
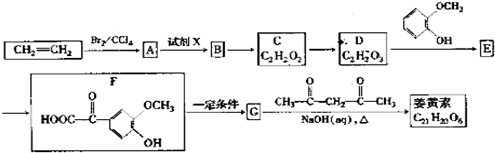

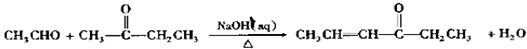

 +OHCCOOH��
+OHCCOOH��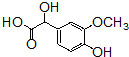 ���䷴Ӧ�����Ǽӳɷ�Ӧ��
���䷴Ӧ�����Ǽӳɷ�Ӧ��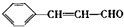 ����д���Ʊ����ȩ�����л���Ľṹ��ʽC6H5CHO��CH3CHO��
����д���Ʊ����ȩ�����л���Ľṹ��ʽC6H5CHO��CH3CHO��
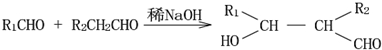
 ��
�� ��
��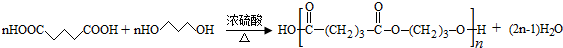 ��
��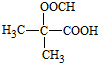 ��
�� ��E��Ԫ�����ڱ��е�λ�õ�������IA�壮
��E��Ԫ�����ڱ��е�λ�õ�������IA�壮