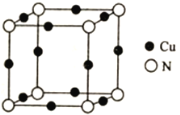
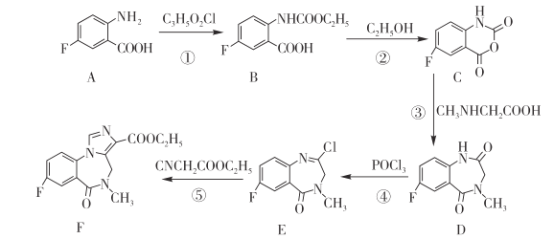
题目内容
【题目】甲基丙烯酸甲酯是合成有机玻璃的单体。旧|法合成的反应是:
①(CH3)2C=O+HCN→(CH3)2C(OH)CN
②(CH3)2C(OH)CN+CH3OH+H2SO4→CH2=C(CH3)COOCH3+NH4HSO4
新法合成的反应是:③CH3C≡CH+CO+CH3OH![]() CH2=C(CH3)COOCH3
CH2=C(CH3)COOCH3
下列说法不正确的是
A.甲基丙烯酸甲酯有两种官能团B.反应③符合绿色化学思想
C.反应②中CH3OH是CH2OHCH2OH(乙二醇)的同系物D.反应①是加成反应
【答案】C
【解析】
A.根据甲基丙烯酸甲酯的结构简式为CH2=C(CH3)COOCH3,该结构中有碳碳双键和酯基两种官能团,故A正确;
B.该反应只有一种目标产物,所以符合绿色化学思想,故B正确;
C.甲醇中含有1个-OH,乙二醇含有2个-OH,二者结构不相似,所以不属于同系物,故C错误;
D.含有碳碳双键(或三键)或碳氧双键的有机物,在某种条件下,双键(三键)中的一条共价键断开,然后在原来形成双键的两个原子上分别连上两个外来的原子或原子团的反应,叫做加成反应,所以反应①符合加成反应,故D正确;
答案为C。

 快乐小博士巩固与提高系列答案
快乐小博士巩固与提高系列答案【题目】运用化学反应原理研究碳、氮、硫的单质及其化合物的反应对缓解环境污染、能源危机具有重要意义。
(1)CO还原NO的反应为2CO(g)+2NO(g)![]() 2CO2(g)+N2(g)H=-746kJ·mol-1。
2CO2(g)+N2(g)H=-746kJ·mol-1。
写出两条有利于提高NO平衡转化率的措施______________、______________。
(2)用焦炭还原NO的反应为:2NO(g)+C(s)![]() N2(g)+CO2(g)
N2(g)+CO2(g)
H。恒容恒温条件下,向体积相同的甲、乙、丙三个容器中分别加入足量的焦炭和一定量的NO,测得各容器中NO的物质的量[n(NO)]随反应时间(t)的变化情况如表所示:
t/min n(NO)/mol 容器 | 0 | 40 | 80 | 120 | 160 |
甲/400℃ | 2.00 | 1.5 | 1.10 | 0.80 | 0.80 |
乙/400℃ | 1.00 | 0.80 | 0.65 | 0.53 | 0.45 |
丙/T℃ | 2.00 | 1.45 | 1.00 | 1.00 | 1.00 |
①H______________0(填“>”或“<”);
②乙容器在160min时,v正_________v逆(填“>”、“<”或“=”)。
(3)某温度下,向体积为2L的恒容真空容器中通入2.0molNO2,发生反应:2NO2(g)![]() N2O4(g)H=-57.0kJ·mol-1,已知:v正(NO2)=k1·c2(NO2),v逆(N2O4)=k2·c(N2O4),其中k1、k2为速率常数。测得NO2的体积分数[x(NO2)]与反应时间(t)的关系如表:
N2O4(g)H=-57.0kJ·mol-1,已知:v正(NO2)=k1·c2(NO2),v逆(N2O4)=k2·c(N2O4),其中k1、k2为速率常数。测得NO2的体积分数[x(NO2)]与反应时间(t)的关系如表:
t/min | 0 | 20 | 40 | 60 | 80 |
x(NO2) | 1.0 | 0.75 | 0.52 | 0.50 | 0.50 |
①![]() 的数值为______________;
的数值为______________;
②已知速率常数k随温度升高而增大,则升高温度后k1增大的倍数___________k2增大的倍数(填“>”、“<”或“=”)。
(4)用间接电化学法除去NO的过程,如图所示:
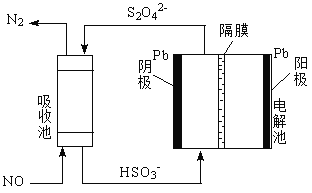
①已知电解池的阴极室中溶液的pH在4~7之间,写出阴极的电极反应式:______________;
②用离子方程式表示吸收池中除去NO的原理:______________。
【题目】一定温度下,20mL0.40mol/LH2O2溶液发生催化分解.不同时刻测得生成O2的体积(已折算为标准状况)如表.
t/min | 0 | 2 | 4 | 6 | 8 | 10 | ||||
V(O2)/mL | 0.0 | 9.9 | 17.2 | 22.4 | 26.5 | 29.9 |
下列叙述不正确的是(溶液体积变化忽略不计)
A.0~6min的平均反应速率:v(H2O2)≈1.67×10-2mol/(Lmin)
B.6~10min的平均反应速率:v(H2O2)<1.67×10-2mol/(Lmin)
C.反应至6min时,c(H2O2)=0.30mol/L
D.反应至6min时,H2O2分解了50%
【题目】下列我国科研成果所涉及材料中,主要成分为同主族元素形成的无机非金属材料的是( )
|
|
|
|
A.4.03米大口径碳化硅反射镜 | B.2022年冬奥会聚氨酯速滑服 | C.能屏蔽电磁波的碳包覆银纳米线 | D.“玉兔二号”钛合金筛网轮 |
A.AB.BC.CD.D