��Ŀ����
6��ij�о���ѧϰС�齫һ��Ũ��Na2C03��Һ����CuS04��Һ�еõ���ɫ��������ͬѧ��Ϊ���߷�Ӧ����ֻ��CuC03һ�ֳ�����
��ͬѧ��Ϊ��������ٽ�ˮ�ⷴӦ������Cu��OH��2һ�ֳ�����
��ͬѧ��Ϊ����CuC03����Cu��0H��2���ֳ���������������֪��CuC03��Cu��oH��2�������ᾧˮ��
������ͬѧ�����⣬Na2C03��Һ��CuS04��Һ��Ӧ�Ļ�ѧ��Ӧ����ʽΪNa2CO3+CuSO4+H2O=Cu��OH��2��+CO2��+Na2SO4����̽��������ɷ�ǰ���뽫��������Һ�з��벢�������������Ϊ�ٹ��ˢ�ϴ�Ӣ۸��
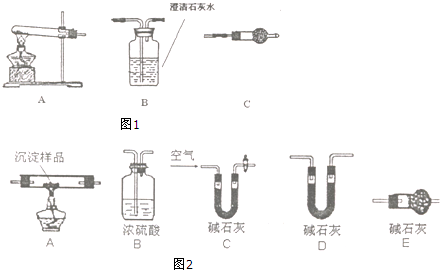
������ͼ1��ʾװ�ã�ѡ���Ҫ���Լ�������̽��������ijɷ֣�
��1����װ������˳��ΪA��C��B��
��2��װ��C��װ���Լ�����������ˮ����ͭ�� l
��3����֤������������CuC03��ʵ��������װ��B�г���ʯ��ˮ����ǣ�
����CuC03��Cu��OH��2���߶��У���ͨ��������ʾװ�ã�ͼ2�����ж����������ⶨ����ɣ�
��1��װ��C�м�ʯ�ҵ����������տ����е�H2O ������CO2��ʵ�鿪ʼʱ��ʵ�����ʱ��Ҫͨ������ģ����������÷ֱ��ǿ�ʼʱͨ�봦�����Ŀ������Խ�װ����ԭ�еĺ�H2O ������CO2�Ŀ���������ʱͨ�˴������Ŀ������Խ�װ����������H2O ������CO2�ϳ�
��2����������Ʒ������Ϊm�ˣ�װ��B����������n�ˣ����������CuC03����������Ϊ��1-$\frac{49n}{9m}$����100%��
���� ������ͬѧ�����⣬Na2C03��Һ��CuS04��Һ��Ӧ�Ƿ�����˫ˮ�ⷴӦ����������ͭ�Ͷ�����̼��
�����ü��ȵķ������飬������ͭ��̼��ͭ���ȷֽ�õ�����ͭ��ˮ�Ͷ�����̼������������ͭ������ˮ����ͭ���飬����̼��ͭ���ó����ʯ��ˮ��������Ķ�����̼������ʯ��ˮ�����˵������CuCO3��
��1��ʵ�鿪ʼʱװ�õĿ����л���ˮ�����Ͷ�����̼�������ų�������װ�����ջ�Լ����������ϴ�����ʿ�ʼʱ���ó�ȥˮ�Ͷ�����̼�Ŀ�����װ���е�ˮ�����Ͷ�����̼�ų���
ʵ�����������ͭ��̼��ͭ���ȷֽ����ڷ�Ӧװ���в���������̼��ˮ������ͨ���ó�ȥˮ�Ͷ�����̼�Ŀ�����װ���е�ˮ�����Ͷ�����̼�ϳ�������װ����ȫ���գ�
��2��װ��B����������n�ˣ�˵���ֽ�����ngˮ������ˮ���������������������ͭ������������������ȥ������ͭ����������̼��ͭ�����������������������Ķ�����㣮
��� �⣺������ͬѧ�����⣬Na2C03��Һ��CuS04��Һ��Ӧ�Ƿ�����˫ˮ�ⷴӦ����������ͭ�Ͷ�����̼����Ӧ�Ļ�ѧ����ʽΪ��Na2CO3+CuSO4+H2O=Cu��OH��2��+CO2��+Na2SO4��
�ʴ�Ϊ��Na2CO3+CuSO4+H2O=Cu��OH��2��+CO2��+Na2SO4��
��1�����ü��ȵķ������飬������ͭ��̼��ͭ���ȷֽ�õ�����ͭ��ˮ�Ͷ�����̼������������ͭ������ˮ����ͭ���飬����̼��ͭ���ó����ʯ��ˮ��������Ķ�����̼��
�ʴ�Ϊ��A��C��B��
��2��װ��C�����Ƿ���ˮ���ɣ�������ˮ����ͭ���飬����ˮ����ͭ����ɫ˵����ˮ���ɣ���֤��������������ͭ���ɣ������������������ͭ��
�ʴ�Ϊ����ˮ����ͭ��
��3���ó����ʯ��ˮ�����Ƿ����������̼��װ��B�г���ʯ��ˮ����ǣ�˵�����ɶ�����̼����˵������CuCO3��
�ʴ�Ϊ��װ��B�г���ʯ��ˮ����ǣ�
��1��ʵ�鿪ʼʱװ�õĿ����л���ˮ�����Ͷ�����̼�������ų�������װ�����ջ�Լ����������ϴ�����ʿ�ʼʱ���ó�ȥˮ�Ͷ�����̼�Ŀ�����װ���е�ˮ�����Ͷ�����̼�ų�����װ��C�м�ʯ�ҵ����������տ����е�H2O������CO2��
������ͭ��̼��ͭ���ȷֽ����ڷ�Ӧװ���в���������̼��ˮ������ͨ���ó�ȥˮ�Ͷ�����̼�Ŀ�����װ���е�ˮ�����Ͷ�����̼�ϳ�������װ����ȫ���գ���ֹӰ��ⶨ�����
�ʴ�Ϊ�����տ����е�H2O ������CO2����ʼʱͨ�봦�����Ŀ������Խ�װ����ԭ�еĺ�H2O ������CO2�Ŀ���������ʱͨ�˴������Ŀ������Խ�װ����������H2O ������CO2�ϳ���
��2��װ��B����������n�ˣ�˵���ֽ�����ngˮ��ˮ�����ʵ���Ϊ$\frac{n}{18}$mol��������Ԫ���غ��֪������ͭ�����ʵ���Ϊ$\frac{n}{18}$mol����������ͭ������Ϊ$\frac{n}{18}$mol��98g/mol=$\frac{49n}{9}$g��������CuCO3����������Ϊ$\frac{m-\frac{49n}{9}}{m}$��100%=��1-$\frac{49n}{9m}$����100%��
�ʴ�Ϊ����1-$\frac{49n}{9m}$����100%��
���� ���⿼���ʵ�鷽�������װ�õ����⡢ʵ�������������ѧ����ȣ��Ѷ��еȣ�����ʵ��ԭ���ǽ���Ĺؼ����Ƕ�֪ʶ���ۺ����ã���Ҫѧ���߱���ʵ�Ļ���֪ʶ������֪ʶ�������⡢��������������

 ӥ�ɽ̸��νӽ̲ĺӱ�����������ϵ�д�
ӥ�ɽ̸��νӽ̲ĺӱ�����������ϵ�д� ���������ν�ϵ�д�
���������ν�ϵ�д�| A�� | �����¶� | B�� | �������������Сһ�� | ||
| C�� | ����C���� | D�� | �����¶� |
| A�� | KNO3��NaCl��--�ᾧ | B�� | ˮ���ƾ���--��Һ | ||
| C�� | Cu��Fe��--�����������ᣬ���� | D�� | CO2��HCl��--����̼��������Һ��ϴ�� |
| A�� | �NaOH��KOH��Ba��OH��2��Na2CO3 | |
| B�� | �����CO2��NO��SO2��H2O | |
| C�� | ��Σ�NH4Cl��NH4NO3����NH4��2SO4��NH3•H2O | |
| D�� | ���������Na2O��CaO��Mn2O7��Al2O3 |
| A�� | 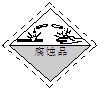 | B�� |  | C�� |  | D�� |  |
 ��
��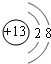 ����д��BԪ�صĵ���������������Һ��Ӧ�����ӷ���ʽ2Al+2OH-+2H2O=2AlO2-+3H2����
����д��BԪ�صĵ���������������Һ��Ӧ�����ӷ���ʽ2Al+2OH-+2H2O=2AlO2-+3H2����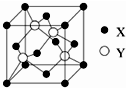 Ԫ��X λ�ڵ������ڣ����̬ԭ�ӵ��ڲ���ȫ���������ӣ�������������Ϊ2��Ԫ��Y��̬ԭ�ӵ�3p �������4�����ӣ�Ԫ��Z ��ԭ�����������������ڲ��3����
Ԫ��X λ�ڵ������ڣ����̬ԭ�ӵ��ڲ���ȫ���������ӣ�������������Ϊ2��Ԫ��Y��̬ԭ�ӵ�3p �������4�����ӣ�Ԫ��Z ��ԭ�����������������ڲ��3����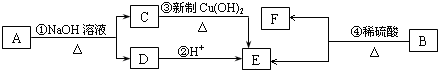
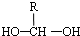 $\stackrel{-H_{2}O}{��}$
$\stackrel{-H_{2}O}{��}$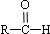 ��R������������
��R������������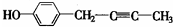 ��
��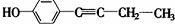 ��
��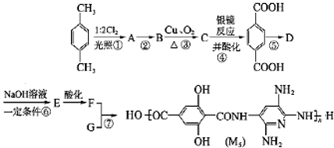
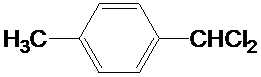 ��д�ṹ��ʽ��
��д�ṹ��ʽ��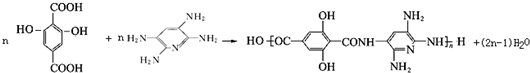 ��
��