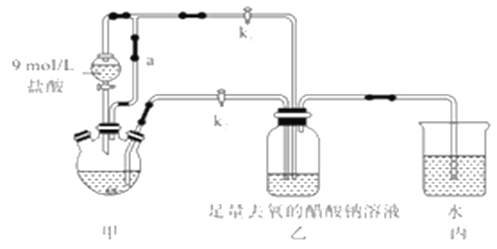
��Ŀ����
����Ŀ��ij��θҩ����Ч�ɷ�Ϊ̼��ƣ��ⶨ����̼��ƺ����IJ������£����ҩƬ�е������ɷֲ���������������Ʒ�Ӧ����
������1.60mol/Lϡ�����1.60mol/LNaOH��Һ��
����һ��������ҩƬ��1.60g���м���20.00mL����ˮ��
����1.60mol/LNaOH��Һ�к�����ϡ���ᣬ��¼�����ĵ�NaOH��Һ�������
�ܼ���25.00mL1.60mol/Lϡ���ᡣ
��ش��������⣺
��1����ȷ�IJ���˳����__________________________������ţ�
��2���ⶨ�����з�����Ӧ�����ӷ���ʽΪ___________________________________��___________________________��
��3���òⶨʵ�鹲������4�Ρ�4�βⶨ�����ĵ�NaOH��Һ��������£�
�ⶨ���� | ��1�� | ��2�� | ��3�� | ��4�� |
V��NaOH(aq)��/mL | 13.00 | 12.90 | 13.10 | 13.00 |
ʵ��������50mL��100mL��250mL��500mL4�ֹ�������ƿ��������NaOH��ҺӦѡ�õ�����ƿ�Ĺ��Ϊ___________��
��4������������NaOH��Һʱ��Ҫ����Ҫ������������ƽ���ձ���________________��
�ڸ�ʵ��ĵ�һ���Ǽ���,��ȡ_____gNaOH���塣
�����ձ���ȡNaOH����ʱ,���в����������ȷ˳����__________________(��ͬ�IJ������ظ�ʹ��)��
a.������ƽ��� b.������Ż������ c.�����벦����̶ȴ�
d.��ȡС�ձ������� e.��ȡС�ձ���NaOH������ f.��¼����������
�ܳ�ȡNaOH����ʱ,�������������ƽ����,��NaOH�����������,������1.4�Ŀ̶��ߴ�����ʵ�ʳƵõ�NaOH����Ϊ____________g��
��д�����в����������Ƶ���ҺŨ����ɵ�Ӱ��(�ƫ��ƫС�����䡱)��
a.����ʱ������NaOH������������λ�õߵ���______________��
b.δϴ���ձ�����������______________��
c.����ҡ�Ⱥ���Һ����ڿ̶��ߣ����¼�ˮ���̶��ߣ�______________��
��ȡ50mL��������ȷ��NaOH��Һ����ˮϡ����100mL������NaOH��Һ�����ʵ���Ũ��Ϊ________________��
���𰸡��٢ڢܢۻ�ڢ٢ܢ� ![]()
![]() 100mL ������ƿ(100mL)����Ͳ��ҩ�ס�����������ͷ�ι� 6.4 adfefbc 3.6 a.ƫС b.ƫС c.ƫС 0.80mol/L
100mL ������ƿ(100mL)����Ͳ��ҩ�ס�����������ͷ�ι� 6.4 adfefbc 3.6 a.ƫС b.ƫС c.ƫС 0.80mol/L
��������
����ʵ�鲽������жϲ���˳��������Һ���ܽ���Ʒ���������ƵĹ���������Һ�ܽ�̼��ƣ�ʣ�������������������Һ�ζ������㷴Ӧ�����ᣬͨ��̼��ƺ����ᷴӦ������ϵ���㺬������������������д��ط�Ӧ����ʽ������������Һԭ����������������
��1������ʵ�鲽������жϲ���˳��������Һ���ܽ���Ʒ���������ƵĹ���������Һ�ܽ�̼��ƣ�ʣ�������������������Һ�ζ������㷴Ӧ�����ᣬͨ��̼��ƺ����ᷴӦ������ϵ���㺬�����ⶨ���̵���ȷ����˳��Ϊ�٢ڢܢۻ�ڢ٢ܢۣ��ʴ�Ϊ���٢ڢܢۻ�ڢ٢ܢۣ�
��2�������з����ķ�ӦΪ̼��ƺ��������������Ȼ��ơ�������̼��ˮ����Ӧ�����ӷ���ʽΪ��CaCO3+2H+=Ca2++CO2��+H2O������������Һ�����ᷴӦ�����Ȼ��ƺ�ˮ����Ӧ�����ӷ���ʽΪ��H++OH=H2O���ʴ�Ϊ��![]() ��
��![]() ��
��
��3��ÿ��ȡ��25.00mLHCl��4����100mL��������NaOH�ζ�ʱ����ҪС��100mL������ѡ�������ƿ���Ϊ100mL���ʴ�Ϊ��100mL��
��4����������Һ���Ƶ�ԭ�������ã���������NaOH��Һʱ��Ҫ����Ҫ������������ƽ���ձ�������ƿ(100mL)����Ͳ��ҩ�ס�����������ͷ�ιܣ��ʴ�Ϊ������ƿ(100mL)����Ͳ��ҩ�ס�����������ͷ�ιܣ�
��m(NaOH)=40g/mol��1.60mol/L��0.1L= 6.4g�����Գ�ȡ6.4gNaOH���壬�ʴ�Ϊ�� 6.4��
�����ձ���ȡNaOH����ʱ������˳��Ϊ��a.������ƽ��㡢d.��ȡС�ձ���������f.��¼���������ݡ�e.��ȡС�ձ���NaOH��������f.��¼���������ݡ�b.������Ż�����У�
�ʴ�Ϊ��adfefbc��
������Ŵ�λ��ʱ����Ʒ������Ӧ���������������ȥ��������������������Ϊ6.4g-1.4g=5g������ʵ������Ϊ5g-1.4g=3.6g���ʴ�Ϊ��3.6g��
��a.����ʱ������NaOH������������λ�õߵ�����Ŵ�λ��ʱ����Ʒ������Ӧ���������������ȥ�����������������Һ������ƫ�٣�����Ũ��ƫС��
b.δϴ���ձ���������������û����ȫת�Ƶ������У�����Ũ��ƫС��
c.����ҡ�Ⱥ���Һ����ڿ̶��ߣ����¼�ˮ���̶��ߣ�ʹ��Ũ��ƫС���ʴ�Ϊ��ƫС��ƫС��c.ƫС��
����Һϡ�����У����ʱ��ֲ��䣬��c1V1=c2V2����50mL��1.60mol/L=100mL��c2��c2=0.80mol/L���ʴ�Ϊ��0.80mol/L��

 ��ʦָ����ĩ��̾�ϵ�д�
��ʦָ����ĩ��̾�ϵ�д�����Ŀ�����и������ʣ����ܰ���ͼ(��������ʾ��Ӧһ�����)��ϵת������
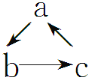
ѡ���� | a | b | c |
A | Al2O3 | NaAlO2 | Al(OH)3 |
B | Al | Al(OH)3 | Al2O3 |
C | AlCl3 | Al(OH)3 | NaAlO2 |
D | MgCl2 | Mg(OH)2 | MgO |
A. AB. BC. CD. D
����Ŀ��ijС���Ի�ͭ��(��Ҫ�ɷ�Ϊ CuFeS2��������SiO2������)Ϊԭ���Ʊ�ͭ������ƷCuAlO2��һ�ֹ������£�

��֪�ٹ���1������Һ�к����������У�Cu2+��Fe2+��Fe3+������1����Ҫ�ɷ���SiO2��S��
��Cu(OH)2+4NH3��H2O=[Cu(NH3)4]2++2OH-+4H2O��
(1)�Ӳ�Ʒ���ȡ�������������ȽǶȿ��ǣ��Լ�A��B����������__________(�����)
a | b | c | d | |
A | HNO3 | NaClO | H2O2 (�ữ) | H2O2 (�ữ) |
B | Cu(OH)2 | NaOH | ��ˮ | Cu(OH)2 CO3 |
(2)������Ļ�ѧʽΪNH4Al(SO4)2��12H2O����Ϊ�Ʊ�ͭ�Ļ�����Ʒ�ṩ��Դ���������Һ��NH4+��A13+��H+��OH-��SO42��Ũ�ȴ�С����Ϊ_______________��
(3)����3�õ�A1(OH)3��Cu(OH)2��д����������������ѧ��Ӧ�ķ���ʽ��_________________��
(4)��λʱ��������ȡ��Cu2+�İٷ���(��Ϊ������)����ҺŨ�ȡ��¶ȹ�ϵ����ͼ��ʾ��
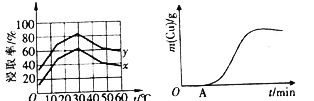
��20��ʱ��1L��Һ����Ч��ײ�ܴ�����x_________y(����>����<������=��)����ͬŨ���£��¶ȸ���30�棬�������������͵���Ҫԭ����_________________��
(5)��ͭ��ʯīΪ�缫�����������1��������Һ�Ʊ�ͭ��ͭ��������ͨ��ʱ���ϵ����ͼ��ʾ��д��OA�������ķ�Ӧ����ʽ��___________________________��
(6)�����£�Ksp[Fe(OH)3]=4.0��10-38�������Լ�B����pH=3ʱc(Fe3+)=_________mol/L