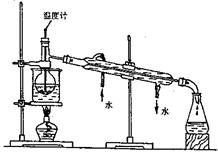
��Ŀ����
���к�������NaCl��Na2SO4��Na2CO3�����ʵ�NaNO3��Һ��ѡ���ʵ����Լ���ȥ���ʣ��õ�������NaNO3���壬ʵ����������ͼ��ʾ��
��1������A����Ҫ�ɷ��� �� ���ѧʽ����
��2�����з�Ӧ�����ӷ���ʽ�� ��
��3���٢ڢ��о����еķ�������� ��
��4�����м��������Na2CO3��Һ��Ŀ���� ��
��5����Һ3�����������Եõ�NaNO3���壬��Һ3�п϶����е������� ��Ϊ�˳�ȥ���ʣ�������Һ3�м��������� ��
��6��ʵ����������ʵ���õ�NaNO3��������500 mL 0.40 mol/L NaNO3��Һ��
��������Һʱ���������²�����a�����ݣ�b�����㣻c���ܽ⣻d��ҡ�ȣ�e��ת�ƣ�f��ϴ�ӣ�j����������ȡNaNO3����������� g�����ղ���˳��4���� ������ţ���
��ijͬѧת����Һ�IJ�����ͼ��ʾ��ͼ���������������ձ��� ����ͬѧ�����еĴ����� ��
��1��BaSO4 BaCO3
��2��Ag++ Cl��="=" AgCl��
��3������
��4����ȥBa2+��Ag+
��5��Na2CO3��CO32�� ϡ����
��6����17.0 e ��500mL����ƿ δ�ò���������
���������������1���������ʵ�NaNO3��Һ��Һ�м��������Ba(NO3)2,������Ӧ��Na2SO4+ Ba(NO3)2= BaSO4��+ 2NaNO3��Na2CO3+ Ba(NO3)2= BaCO3��+ 2NaNO3����A����Ҫ�ɷ���BaSO4��BaCO3����2����Һ1�к���NaCl��NaNO3��������Ba(NO3)2�������м��������Ag NO3��Һ��������Ӧ��NaCl +Ag NO3= AgCl��+ NaNO3�����ӷ���ʽΪ��Ag++ Cl��="=" AgCl������3���٢ڢ��ж��Ƿ��������Թ�����Һ��IJ����������еķ�������ǹ��ˡ���4������Һ2�м��������Na2CO3��Һ��������Ӧ��Ba(NO3)2+ Na2CO3= BaCO3��+ 2NaNO3��2AgNO3+ Na2CO3= Ag2CO3��+ 2NaNO3.���Ԣ��м��������Na2CO3��Һ��Ŀ���dz�ȥBa2+��Ag+����5�����ڢۼ��������Na2CO3��Һ������3�����������Եõ�NaNO3���壬��Һ3�п϶����е�������Na2CO3��Ϊ�˳�ȥ���ʣ�������Һ3�м���������ϡ���ᡣ��6����������Һʱ����ȡNaNO3�����������0.5L ��0.40 mol/L��85g/mol=17.0g.���ղ���˳��4����e��ת��.��ijͬѧת����Һ�IJ�������ͼ��ʾ��ͼ���������������ձ���500mL����ƿ;��ͬѧ�����еĴ�����δ�ò�����������
���㣺�������ʵij�ȥ�����ӷ���ʽ����д�����ʵ���Ũ�ȵ���Һ�����Ʋ��衢������ʹ�õ�֪ʶ��

������㷺Ӧ������ҩ�ͻ�����ҵ��ijͬѧ�����üױ���������Ӧ�Ʊ������ᡣ��Ӧԭ��: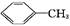 +2KMnO4
+2KMnO4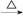
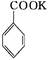 +KOH+
+KOH+
2MnO2��+H2O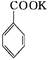 +HCl
+HCl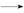
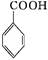 +KCl
+KCl
ʵ�鷽��:һ�����ļױ���KMnO4��Һ��100 �淴Ӧһ��ʱ���ֹͣ��Ӧ,���������̷����������ͻ���δ��Ӧ�ļױ���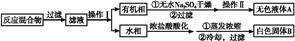
��֪:�����������122,�۵�122.4 ��,��25 ���95 ��ʱ�ܽ�ȷֱ�Ϊ0.3 g��6.9 g;���������л���һ�㶼�й̶��۵㡣
(1)��������������,����������������
(2)��ɫҺ��A����������,���Լ���A���Լ�����������,�������� ��
(3)�ⶨ��ɫ����B���۵�,��������115 �濪ʼ�ۻ�,�ﵽ130 ��ʱ�����������ۡ���ͬѧ�Ʋ��ɫ����B�DZ�������KCl�Ļ����,��������·��������ᴿ�ͼ���,ʵ���������Ʋ���ȷ������ɱ������ݡ�
| ��� | ʵ�鷽�� | ʵ������ | ���� |
| �� | ����ɫ����B����ˮ��,�����ܽ�,������ | �õ���ɫ�������ɫ��Һ | |
| �� | ȡ������Һ���Թ���,�������� | ���ɰ�ɫ���� | ��Һ��Cl- |
| �� | �����ɫ����,������ | ������ | ��ɫ���� �DZ����� |
(4)���Ȳⶨ:��ȡ1.220 g��Ʒ,���100 mL�״���Һ,��ȡ25.00 mL��Һ,�ζ�,����KOH�����ʵ���Ϊ2.40��10-3 mol����Ʒ�б��������������ļ������ʽΪ��������,������Ϊ��������(������λ��Ч����)��
1�������������������ϣ�Ҳ�ɺϳ��������ϡ�ʵ�����Ʊ�1�����������Ĺ������£� 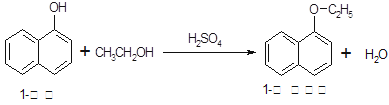
��֪����������ʵ���������
| ���� | ��Է� ������ | ״̬ | �۵�(��) | �е�(��) | �ܽ�� | |
| ˮ | �Ҵ� | |||||
| 1������ | 144 | ��ɫ���ɫ���νᾧ���ĩ | 96�� | 278�� | ����ˮ | �������Ҵ� |
| 1���������� | 172 | ��ɫҺ�� | 5��5�� | 267�� | ������ˮ | �������Ҵ� |
| �Ҵ� | 46 | ��ɫҺ�� | -114��1�� | 78��5�� | ����Ȼ��� | |
��1�����ӵ������뱽�����ƣ������ŵı�����ζ�������ڿ������ױ�����Ϊ�ۺ�ɫ��
��ش��������⣺
��1����72g1����������100mL��ˮ�Ҵ��У�����5mLŨ�����ϡ������Һ������ͼ��ʾ�������м��ȳ�ַ�Ӧ��ʵ����ʹ�ù����Ҵ���ԭ���� ��

��2����Ӧ����������ƿ�е�Һ�嵹����ˮ�У��������õ��л��㡣Ϊ�ᴿ�����������IJ�������������ˮϴ����Һ������10%��NaOH��Һ��ϴ����Һ��������ˮ�Ȼ��Ƹ��ﲢ���ˡ���ȷ��˳���� (�����)��
A���ۢڢܢ� B���٢ڢۢ� C���ڢ٢ۢ�
��3��ʵ����1�����������IJ����뷴Ӧʱ�䡢�¶ȵı仯��ͼ��ʾ��ʱ���ӳ����¶����ߣ�1�����������IJ����½���ԭ������� ��
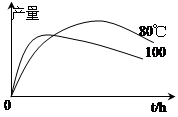
��4���ᴿ�IJ�Ʒ���ⶨΪ43g����ʵ����1�����������IJ���Ϊ ��
CuSO4��Һ��Na2CO3��Һ��ϲ�������ɫ������������ij��ȤС��Գ�����ɵ�̽����
��������衿
����1������ΪCu (OH)2
����2������Ϊ
����3������Ϊ��ʽ̼��ͭ[��ѧʽ�ɱ�ʾΪnCuCO3��mCu (OH)2]
���������ϡ���������һ�ֳ������Ⱦ��ֽ⣨����������ᾧˮ����
������̽����
����1������������Һ���ˣ�������ˮϴ�ӣ�������ˮ�Ҵ�ϴ�ӣ���ɣ�
����2����ͬѧȡһ�������壬�����������õ�����װ�ã��г�����δ���������ж���ʵ�飻
��1������Ӧ��A������ɫ�����ڣ�C������������֤������ ������
��2����ͬѧ��ΪֻҪ����ͼ��Bװ�õ��Լ���������ij�Լ������֤�������м��裬���Լ��� ������ţ���
a��Ũ���� b����ˮCuSO4 c����ʯ�� d��P2O5
��3����ͬѧ����B�Լ�����֤����3������ʵ�������� ��
������̽����
��4����ͬѧ��һ��̽������3�й������ɣ�
����ͬѧ���һЩ������20������ݣ����±�����C�еij���ʯ��ˮ��ΪBa(OH)2��Һ����ԭ���� ��˫ѡ������ţ�
| �ܽ��(S)/g | �ܶȻ�(Ksp) | Ħ������(M)/g��mol��1 | |||
| Ca(OH)2 | Ba(OH)2 | CaCO3 | BaCO3 | CaCO3 | BaCO3 |
| 0.16 | 3.89 | 2.9��10-9 | 2.6��10-9 | 100 | 197 |
b��Ba(OH)2Ϊǿ�Ca(OH)2Ϊ����
c�����յ���CO2���ɵ�BaCO3����������CaCO3���������С
d����ͬ�����£�CaCO3���ܽ�����Դ���BaCO3
������ȡ����ɫ��������Ϊ54.2 g��ʵ�������װ��B����������5.4 g��C�еIJ�������������Ϊ39.4 g���������ɫ����Ļ�ѧʽΪ ��