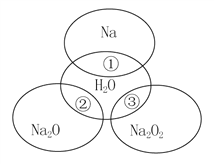
��Ŀ����
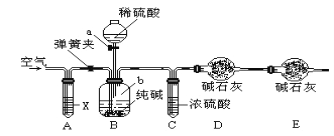
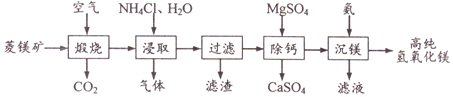
����Ŀ��һ������þ��(��Ҫ�ɷ�MgCO3��CaCO3��FeCO3��SiO2)�Ʊ��ߴ�������þ�Ĺ�������:
��1����������ʱFeCO3������Ӧ�ķ���ʽΪ_____��
��2������ȡ���¶�100�����ң��ò��躬þ��������뷴Ӧ�Ļ�ѧ����ʽΪ______��
��3������ȡ��ʱNH4Cl������Mg2+�����ʵ�Ӱ����������ͼ��ʾ�������й�˵����ȷ��____(�����)��
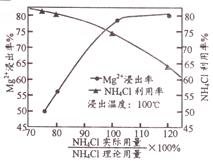
A.����ȡ��ʱ�ʵ����转��
B.�ó���������20%��NH4Cl���н�ȡ
C.����NH4Cl�Խ�ȡ����������ж��ν�ȡ�������ν�ȡҺ�ϲ�
D.�����¶Ȳ���100���ɴٽ�NH4Cl��ˮ���NH3���ݳ�
��4�������������п�ѭ�����õ�������______��
��5����Ҫ�õ��ߴ�Mg(OH)2���������þ���������õĹ������ϴ�ӣ����������ϴ���ķ�����_______��
��6��25��ʱ��Ksp(CaSO4)=7.2��10-5��ʵ������������ǰ����Һ��c(Ca2+)�ֱ�Ϊ2.8g/L��0.8g/L,��1L��Һ���������MgSO4���������Ϊ____g��
���𰸡� 4FeCO3+O2![]() 2Fe2O3+4CO2 MgO+2NH4Cl
2Fe2O3+4CO2 MgO+2NH4Cl![]() MgCl2+2NH3+H2O ACD NH4Cl��NH3 ȡ���һ��ϴ����Һ�����������ữ��BaCl2��Һ�����ް�ɫ�������ɣ�����ϴ�� 6.4g
MgCl2+2NH3+H2O ACD NH4Cl��NH3 ȡ���һ��ϴ����Һ�����������ữ��BaCl2��Һ�����ް�ɫ�������ɣ�����ϴ�� 6.4g
����������1����þ���к���FeCO3����������ʱFeCO3������е���������������ԭ��Ӧ����Ӧ�ķ���ʽΪ��4FeCO3��O2![]() 2Fe2O3��4CO2����Ϊ��4FeCO3��O2
2Fe2O3��4CO2������4FeCO3��O2![]() 2Fe2O3��4CO2
2Fe2O3��4CO2
��2������ȡ��ʱ�����¶�100 �����ң���ʱ����������������MgO��NH4Cl��ˮ��Һ������Ӧ����Ӧ�Ļ�ѧ����ʽΪ��MgO��2NH4Cl![]() MgCl2��2NH3����H2O����Ϊ��MgO��2NH4Cl
MgCl2��2NH3����H2O������MgO��2NH4Cl![]() MgCl2��2NH3����H2O
MgCl2��2NH3����H2O
��3��A. ����ȡ��ʱ�ʵ����转������Ӵ������������Mg2��������A��ȷ��B. ��ͼ���֪�����ó���������20%��NH4Cl���н�ȡʱ��NH4Cl�������ʽϵͣ�B������C.��NH4Cl�Խ�ȡ����������ж��ν�ȡ�������ν�ȡҺ�ϲ��������Mg2�������ʣ�C��ȷ��D. �����¶Ȳ���100 ��ɴٽ�NH4Cl��ˮ�⣬����������ǿ��NH3���ݳ���������MgO���ܽ⣬�Ӷ����Mg2�������ʣ�D��ȷ����ѡACD.
(4) ����þ���Ʊ��ߴ�������þ�Ĺ����������õ�NH4Cl��NH3����˱�����������NH4Cl��NH3��ѭ�����á���Ϊ��NH4Cl��NH3
��5������Mg(OH)2������ϴ����ֻҪ����ϴ��Һ��û��Cl-��������˼��������ϴ���ķ�����ȡ���һ��ϴ����Һ�����������ữ��BaCl2��Һ�����ް�ɫ�������ɣ�����ϴ������Ϊ��ȡ���һ��ϴ����Һ�����������ữ��BaCl2��Һ�����ް�ɫ�������ɣ�����ϴ����
��6����Һ��c(SO42-)=![]() =
=![]() =3.6
=3.6![]() mol��L��1, 1 L��Һ���ɵ�CaSO4Ϊ
mol��L��1, 1 L��Һ���ɵ�CaSO4Ϊ![]() =0.05mol��������ҪMgSO4���������Ϊ3.6
=0.05mol��������ҪMgSO4���������Ϊ3.6![]() mol��L��1��1 L��120g/mol+0.05 mol��120g/mol=6.4g����Ϊ:6.4
mol��L��1��1 L��120g/mol+0.05 mol��120g/mol=6.4g����Ϊ:6.4