��Ŀ����
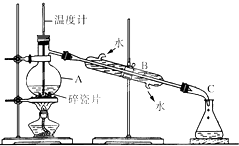
8��ʵ�����������᳧��������Ҫ�ɷ�Ϊ���������P����FeS SiO2�ȣ��Ʊ���������ʽ�������ľۺ�����̷���FeSO4•7H2O�����������ͼ��
��1�����̢��У�FeS��02��H2S04��Ӧ�Ļ�ѧ����ʽΪ4FeS+3O2+6H2SO4=2Fe2��SO4��3+6H2O+4S��
��2�����̢��в�����β����Դ��������Ⱦ����ѡ�������Լ��е�c���գ�
a��ŨH2SO4 b������ˮ c��NaOH��Һ d��Ũ����
��3�����̢��У���Ҫ���˵�����������Fe����������
��4�����̢ܵ�ʵ������������ᾧ
��5�����̢��У�����ҺZ���ȵ�70〜800C��Ŀ���Ǵٽ�Fe3+��ˮ�⣮
��6��ʵ����Ϊ�ⶨ���õ��ľ�����Ʒ����Ԫ�ص�������������������ʵ�飮���÷�����ƽ��ȡ ��Ʒ2.700g���ڽ���Ʒ����������������������Ȼ�����Һ���۹��ˡ�ϴ�ӡ������������ ��������Ϊ3.495g�����þ�����Ҫ�ɷ�Ϊ[Fe��0H��S04]����þ�������Ԫ�ص���������Ϊ31.11%��
���� �����м���ϡ���ᡢͬʱͨ�����������˺õ�����W�к���S��SiO2�������պ�β���к��ж���������ҺX����Fe2��SO4��3��δ��Ӧ�����ᣬ��ҺX�м���Fe�ۣ���Ӧ��ϣ����˵õ���ҺYΪFeSO4��Һ���پ�������Ũ������ȴ�ᾧ�����ˡ�ϴ�ӵȵõ��̷�����ҺX����pH�õ���ҺZ�����ȴٽ�Fe3+��ˮ��õ��������壬�پ�������۳��õ�������
��1�����̢�FeS��O2��H2SO4��Ӧ������������ˮ����
��2��β���к��ж���������Ҫ��NaOH��Һ���գ�
��3����ҺX�к���Fe3+��Ҫ�Ʊ��̷���Ӧ����Feʹ֮��ԭΪFe2+��
��4�����̢�����Һ�еõ����ʾ������ͨ�������ᾧ�õ���
��5�����̢�Ŀ��Ϊ�ٽ���ˮ�⣬����������ˮ��Ľ��У�
��6��������еõ�3.495g����ΪBaSO4����������n=$\frac{m}{M}$����BaSO4�����ʵ���������������غ㣬���Լ������������������ʵ�������Ͼ�����Fe�������������ϵ����Fe�����ʵ������ٸ���m=nM������Ԫ������������������Ԫ������������
��� �⣺�����м���ϡ���ᡢͬʱͨ�����������˺õ�����W�к���S��SiO2�������պ�β���к��ж���������ҺX����Fe2��SO4��3��δ��Ӧ�����ᣬ��ҺX�м���Fe�ۣ���Ӧ��ϣ����˵õ���ҺYΪFeSO4��Һ���پ�������Ũ������ȴ�ᾧ�����ˡ�ϴ�ӵȵõ��̷�����ҺX����pH�õ���ҺZ�����ȴٽ�Fe3+��ˮ��õ��������壬�پ�������۳��õ�������
��1����Ӧ��ΪFeS��O2��H2SO4����������S�����������غ㻹Ӧ��Fe2��SO4��3��H2O����Ӧ�Ļ�ѧ����ʽΪ4FeS+3O2+6H2SO4=2Fe2��SO4��3+6H2O+4S��
�ʴ�Ϊ��4FeS+3O2+6H2SO4=2Fe2��SO4��3+6H2O+4S��
��2��β���к��ж���������Ҫ��NaOH��Һ���գ���ѡc��
��3����ҺX�к���Fe3+��Ҫ�Ʊ��̷���Ӧ����Feʹ֮��ԭΪFe2+��������������Fe2+�����Ҳ������µ����ʣ��ʴ�Ϊ��Fe����������
��4�����̢�����Һ�еõ����ʾ������ͨ�������ᾧ�õ����ʴ�Ϊ�������ᾧ��
��5�����̢�Ŀ��Ϊ�ٽ���ˮ�⣬����������ˮ��Ľ��У��ٽ�Fe3+��ˮ�⣬�ʴ�Ϊ���ٽ�Fe3+��ˮ�⣻
��6�����ˡ�ϴ�ӡ�����������ù�������Ϊ3.495g��ӦΪBaSO4������
n��BaSO4��=$\frac{3.495g}{233g/mol}$=0.015mol��
��n��Fe3+��=n��SO42-��=0.015mol��
m��Fe��=0.015mol��56g/mol=0.84g��
�أ�Fe��=$\frac{0.84g}{2.7g}$��100%=31.11%��
�ʴ�Ϊ��31.11%��
���� ���⿼���Ϊ�ۺϣ��漰���ʺ����IJⶨ���Ʊ�ʵ�鷽������Ƶ�֪ʶ����Ŀ�ѶȽϴ�ע�ⳣ����ѧʵ�����������

 ��������������������ϵ�д�
��������������������ϵ�д�| A�� | ԭ�Ӱ뾶��С��Na��S��O | B�� | ����ǿ����HClO4��HBrO4��HIO4 | ||
| C�� | ����ǿ����Ba��OH��2��Sr��OH��2��Ca��OH��2 | D�� | �ǽ�����ǿ����Cl��Se��Br |
| A�� | Cl2 | B�� | HCl | C�� | SO2 | D�� | CO2 |
| A�� | NH3�ĵ���ʽ�� | |
| B�� | �����ӵĽṹʾ��ͼ��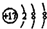 | |
| C�� | CS2�Ľṹʽ��S=C=S | |
| D�� | ԭ�Ӻ�����10�����ӵ���ԭ�ӣ�${\;}_{8}^{18}$O |

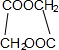 +2H2O��
+2H2O��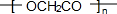 ��
��
 �ṹ���Ƶ��л��ﲻ�ȶ�������������������
�ṹ���Ƶ��л��ﲻ�ȶ�������������������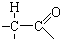
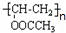 ��
��