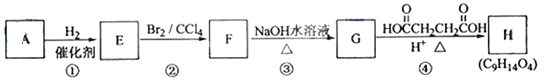
��Ŀ����
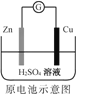
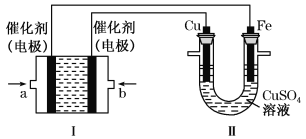
����Ŀ������ͼ��ʾ��װ�â�Ϊ����ȼ�ϵ��(�������ҺΪKOH��Һ)��ͨ��װ�â�ʵ�������϶�ͭ��
��a��Ӧͨ��______(����CH4������O2��)��a���缫�Ϸ����ĵ缫��Ӧʽ��______________��
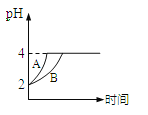
�ڵ�ƽ�����װ�â�����Һ��pH _________ (��д�����������С����������������ͬ)��װ�â���Cu2+�����ʵ���Ũ��____________________��
�۵�ƽ�����װ�â���Һ�е������ӳ���OH-�������________________ ��
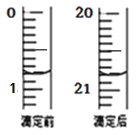
���ڴ˹���������ȫ��Ӧ��װ�â������������仯6.4 g����װ�â������������ļ���__________L(��״����)��
���𰸡�CH4 CH4+10OH--8e-=CO32-+7H2O ��С ���� CO32- 0.56
��������
��II�����ȶ�ͭ����Cu��������Fe��������I��a���缫Ϊ������b���缫Ϊ������������ͨ��ȼ�ϡ�������ͨ��������������ʧ���Ӻ����������ӷ�Ӧ����̼������Ӻ�ˮ��
�ڸ���I������������Ũ�ȱ仯ȷ����ҺpH�仯��II�з�����ƣ��������ܽ��ͭ������������������ͭ��������
��I�л���̼����������ɣ�
�ܸ���ת�Ƶ�����ȼ�����ʧ����������
��װ��II��ʵ�����϶�ͭ����Cu��������Fe��������I��a���缫Ϊ������b���缫Ϊ������������ͨ��ȼ�ϡ�������ͨ��������������a��ͨ��������Ǽ��飻����ʧ���Ӻ����������ӷ�Ӧ����̼������Ӻ�ˮ���缫��ӦΪCH4+10OH--8e-=CO32-+7H2O��b��ͨ�����������缫�Ϸ����ĵ缫��Ӧʽ�ǣ�O2+2H2O+4e-=4OH-��
�ڸ���I�е�ط�ӦΪCH4+2O2+2KOH=K2CO3+3H2O��KOH�μӷ�Ӧ������Һ��KOHŨ�Ƚ��ͣ�������Һ��pH��С��
II�з�����ƣ��������ܽ��ͭ������������������ͭ�������������Һ��ͭ����Ũ�Ȳ��䣻
��I�и�����ӦΪCH4+10OH--8e-=CO32-+7H2O��������Һ�г��˺���OH-�⣬����CO32-��
��n(Cu)=![]() =0.1 mol��Cu��+2�۵Ľ��������Է�Ӧ�����е���ת�Ƶ����ʵ���Ϊ0.1 mol��2=0.2 mol�����ڴ�����·��ת�Ƶ�����ȣ����Է�Ӧ���ļ�������V(CH4)=
=0.1 mol��Cu��+2�۵Ľ��������Է�Ӧ�����е���ת�Ƶ����ʵ���Ϊ0.1 mol��2=0.2 mol�����ڴ�����·��ת�Ƶ�����ȣ����Է�Ӧ���ļ�������V(CH4)=![]() ��22.4 L/mol=0.56 L��
��22.4 L/mol=0.56 L��

 �������Ӧ���⼯ѵϵ�д�
�������Ӧ���⼯ѵϵ�д� �ۺ��Բ�ϵ�д�
�ۺ��Բ�ϵ�д�