��Ŀ����
����Ŀ����֪��1 mol�����еĻ�ѧ����Ҫ����436 kJ��������1 mol�����еĻ�ѧ����Ҫ����498 kJ������������ͼ�е�����ͼ���ش��������⣺
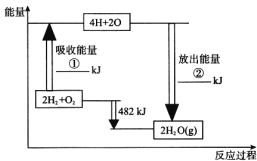
��1���ֱ�д���٢�����ֵ��
��__________����__________��
��2������H2(Xg)�е�1 mol H��O���ų�__________kJ��������
��3����֪��H2O(l)![]() H2O(g)��H��+44 kJ mol1����д����������������ȫȼ������Һ̬ˮ���Ȼ�ѧ����ʽ��___________________________________��
H2O(g)��H��+44 kJ mol1����д����������������ȫȼ������Һ̬ˮ���Ȼ�ѧ����ʽ��___________________________________��
���𰸡���1����1370 ��1 852
��2��463
��3��2H2(g)+ O2(g)![]() 2H2O��l�� ��H=482 kJ mol1
2H2O��l�� ��H=482 kJ mol1
����������1����������Ϊ����2 mol H��H�����ܵ���ֵ��1 mol O=O�����ܵ��ܺͣ�ͬ���ĵ�������������Ϊ4 mol H��2 mol O�γ�2 mol H2O(g)�ͷŵ����������γ�4 mol H��O���ͷŵ���������2���γ�4 mol H��O���ͷŵ�����Ϊ1 852 kJ��������H2(Xg)�е�1 mol H��O���ų�463 kJ��������

��ϰ��ϵ�д�
�����Ŀ

