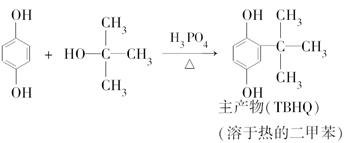
��Ŀ����
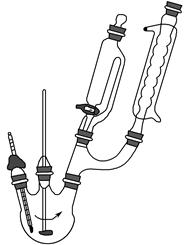
ij�о�С������ͼװ�ý���SO2��FeCl3��Һ��Ӧ�����ʵ�飨�г�װ������ȥ����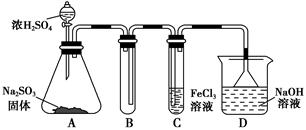
��1���������Ȼ�����Һʱ�����Ȱ��Ȼ��������ܽ���________�У��ټ�ˮϡ�ͣ�����������Ŀ����________�������в���Ҫ��������________��������ȷѡ��ǰ����ĸ����
a��ҩ�ס� b���ձ��� c��ʯ������ d���������� e������
��2��ͨ������SO2ʱ��C�й۲쵽������Ϊ______________________________��
��3��������������С��ͬѧ��ΪSO2��FeCl3��Һ������������ԭ��Ӧ��
��д��SO2��FeCl3��Һ��Ӧ�����ӷ���ʽ��___________________________��
�������ʵ�鷽��������Fe2�����ɣ�__________________________________��
�۸�С��ͬѧ��C�Թܷ�Ӧ�����Һ�м��������ữ��BaCl2��Һ�������ְ�ɫ����������֤����Ӧ������SO42-��������________���������������������������_______________________________________________________________��
��4��Dװ���е���©����������______________________________________��
��5��Ϊ����֤SO2���л�ԭ�ԣ�ʵ���п��Դ���FeCl3���Լ���________��������ȷѡ��ǰ����ĸ����
a��ŨH2SO4 b������KMnO4��Һ
c����ˮ d��NaCl��Һ
��1��Ũ���ᡡ�����Ȼ���ˮ�⡡ce
��2����Һ���ػ�ɫ��Ϊdz��ɫ
��3����2Fe3����SO2��2H2O=SO42-��4H����2Fe2��
��ȡC�з�Ӧ�����Һ�������е���KSCN��Һ������죬�ټ������Ƶ���ˮ����H2O2��Һ�ȣ�����Һ���
�۲�������������Խ��ܽ��SO2����ΪH2SO4������ʵ��
��4��ʹ���������գ���ֹ��Һ����
��5��bc
����

����ȩ��һ�ֻ���ԭ�ϡ�ijʵ��С����������װ�úϳ�����ȩ��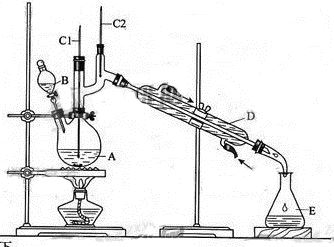
�����ķ�Ӧ���£�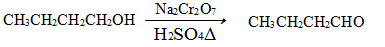
��Ӧ��Ͳ������������б����£�
| | �е�/�� | �ܶ�/(g��cm-3) | ˮ���ܽ��� |
| ������ | 11.72 | 0.8109 | �� |
| ����ȩ | 75.7 | 0.8017 | �� |
ʵ�鲽�����£�
��6.0gNa2Cr2O7����100mL�ձ��У���30mLˮ�ܽ⣬�ٻ�������5mLŨ���ᣬ��������ҺС��ת����B�С���A�м���4.0g�������ͼ�����ʯ�����ȡ�������������ʱ����ʼ�μ�B����Һ���μӹ����б��ַ�Ӧ�¶�Ϊ90��95�棬��E���ռ�90�����µ���֡�������ﵹ���Һ©���У���ȥˮ�㣬�л������������ռ�75��77����֣�����2.0g��
�ش��������⣺
(1)ʵ���У��ܷ�Na2Cr2O7��Һ�ӵ�Ũ�����У�˵������ ��
(2)�����ʯ�������� �������Ⱥ���δ�ӷ�ʯ��Ӧ��ȡ����ȷ������ ��
(3)����װ��ͼ�У�B������������ ��D������������ ��
(4)��Һ©��ʹ��ǰ������еIJ����� (����ȷ�𰸱��)��
a����ʪ b������ c����© d���궨
(5)������ȩ�ֲ�Ʒ���ڷ�Һ©���з�ˮʱ��ˮ�� ��(��ϡ����¡�)
(6)��Ӧ�¶�Ӧ������90��95�棬��ԭ���� ��
(7)��ʵ���У�����ȩ�IJ���Ϊ %��
�����ѳ������л���Ӧ���ܼ���ʵ�����Ʊ������ѵķ�Ӧ����Ҫʵ��װ�����£�
2CH3CH2CH2CH2OH (CH3CH2CH2CH2)2O+H2O
(CH3CH2CH2CH2)2O+H2O
��Ӧ��Ͳ���������������
| | ��Է������� | �е�/�� | �ܶ�/��g/cm3�� | ˮ���ܽ��� |
| ������ | 74 | 117��2 | 0��8109 | �� |
| ������ | 130 | 142��0 | 0��7704 | �������� |
�ٽ�6 mLŨ�����37 g����������һ��˳�����ӵ�A�У����Ӽ�����ʯ��
�ڼ���A�з�ӦҺ��Ѹ��������135�棬ά�ַ�Ӧһ��ʱ�䡣�����ᴿ��
�۴�A��Һ����ȴ���仺������ʢ��70 mLˮ�ķ�Һ�Ҷ��У���ҡ���ã���Һ�ôֲ��
�ֲܴ���������40 mLˮ��20 mL NaOH��Һ��40 mLˮϴ�ӣ���Һ�����Լ3 g��ˮ�Ȼ��ƿ���������һ��ʱ�����ȥ�Ȼ��ơ�
�ݽ������������Ĵֲ�����������ռ���֣��ô���������11 g��
��ش�
��1���������Ũ�����������������˳��Ϊ ��
��2������Aǰ�����ȴ�____���a����b��������B��ͨ��ˮ��
��3������۵�Ŀ���dz���ϴȥ ����ҡ���ã��ֲ���Ӧ�ӷ�Һ©���� ����ϡ����¡����ڷ������
��4������������һ��ˮϴ��Ŀ��Ϊ ��
��5��������У���������ʱӦ�ռ� ����ѡ����ĸ�����ҵ���֡�
A��100�� B��117�� C��135�� D��142��
��6����Ӧ�����л�۲쵽��ˮ�����ռ���Һ�����ʣ��ҷ�Ϊ�������㣬���ŷ�Ӧ�Ľ��У���ˮ����Һ��������������ʱ���ϲ�Һ�������֧���Զ�����A����ˮ�����ϲ�Һ�����Ҫ�ɷ�Ϊ____���²�Һ�����Ҫ�ɷ�Ϊ ��
��7����ʵ���У������ѵIJ���Ϊ ��
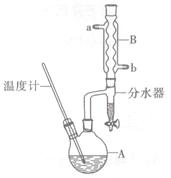
����������ͨ���÷��������������Ʊ����������ı����Ƚ��ȶ������������������ϵ�֧�����۳��̣���ǿ������ʱ�����ն��������Ȼ���ijͬѧ�üױ���������Ӧ�Ʊ������ᡣ��Ӧԭ����
��Ӧ�Լ������������������
| ���� | ��Է������� | ��״ | �۵� | �е� | �ܶ� | �ܽ�� | ||
| ˮ | �Ҵ� | ���� | ||||||
| �ױ� | 92 | ��ɫҺ����ȼ�ӷ� | -95 | 110.6 | 0.8669 | ���� | �� | �� |
| ������ | 122 | ��ɫƬ״����״���� | 122.4 | 248 | 1.2659 | �� | ���� | ���� |

ͼ1��������װ�� ͼ2����װ��
ʵ�鷽����һ�����ļױ���KMnO4��Һ����ͼ1װ���У���100��ʱ����Ӧһ��ʱ�䣬��ֹͣ��Ӧ�����������̷����������ͻ���δ��Ӧ�ļױ���
��1������K������Ϊ ����ɫҺ��A�Ľṹ��ʽΪ ��������Ϊ ��
�����Һ����ɫ��Ҫ�ȼ���������أ�Ȼ���ټ���Ũ�����ữ������������ص�Ŀ���� ��
��3�����й�����������װ����ʹ����ȷ���� ��
A�����˿��Լӿ�����ٶȣ��õ��ϸ���ij���
B����װ�綯������ʱ��������¶˲�����������ƿ�ס��¶ȼƵȽӴ�
C�����˽�����Ϊ��ֹ������Ӧ�ȹر�ˮ��ͷ���ٶϿ����ϵͳ�����ϵͳ������
D����������ˮ���������½��ϳ�
��4����ȥ�����ڱ������еļױ�Ӧ�ȼ��� ����Һ��ˮ���ټ��� ��Ȼ����ˣ����T�ɵõ������ᡣ
��5�����Ȳⶨ����ȡ1.220g��Ʒ�����100ml��Һ��ȡ����25.00ml��Һ�����еζ�������KOH���ʵ���Ϊ2.4��10-3mol����Ʒ�б�������������Ϊ ��
��ǹ�ҵ���Ʊ�Na2S2O3�ķ���֮һ����Ӧԭ��Ϊ��2Na2S+Na2CO3+4SO2=3Na2S2O3+CO2 ���÷�Ӧ��H>0��
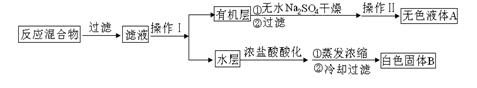
ij�о�С����ʵ��������Ʊ�Na2S2O3��5H2O�������¡�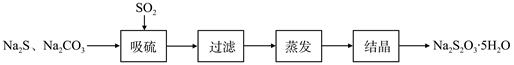
��1������װ����ͼ��ʾ��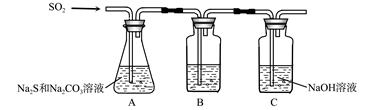
��װ��B�������Ǽ���װ��A��SO2������Ч�ʣ�B���Լ��� ������SO2����Ч�ʵ͵�ʵ��������B����Һ ��
��Ϊ��ʹSO2������������ȫ���ڲ��ı�A����ҺŨ�ȡ�����������£����˼�ʱ���跴Ӧ���⣬���ɲ�ȡ�ĺ�����ʩ�� �� ����д��������
��2�����豾ʵ�����õ�Na2CO3������NaCl��NaOH�����ʵ�鷽�����м��顣������ʱCaCO3������Һ��pH=10.2��
��ѡ�Լ���������ϡ���ᡢAgNO3��Һ��CaCl2��Һ��Ca��NO3��2��Һ����̪��Һ������ˮ��pH�ơ��ձ����Թܡ��ι�
| ��� | ʵ����� | Ԥ������ | ���� |
| �� | ȡ������Ʒ���Թ��У�������������ˮ��������ܽ⣬___________________�� | _______________ | ��Ʒ��NaCl |
| �� | ��ȡ������Ʒ���ձ��У�������������ˮ����ֽ����ܽ⣬___________________�� | _______________ | ��Ʒ��NaOH |
��3��Na2S2O3��Һ�Ƕ���ʵ���еij����Լ����ⶨ��Ũ�ȵĹ������£�ȷ��ȡa g KIO3����ѧʽ����214�����������Һ���������KI�����H2SO4��Һ���μ�ָʾ������Na2S2O3��Һ�ζ����յ㣬����Na2S2O3��Һ�����ΪV mL����c��Na2S2O3����_________mol��L��1����ֻ�г���ʽ���������㣩
��֪��IO3����5I��+6H+== 3I2��3H2O 2S2O32����I2==S4O62����2I��
������㷺Ӧ������ҩ�ͻ�����ҵ��ijͬѧ�����üױ���������Ӧ�Ʊ������ᡣ��Ӧԭ��: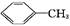 +2KMnO4
+2KMnO4
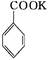 +KOH+
+KOH+
2MnO2��+H2O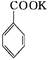 +HCl
+HCl
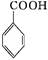 +KCl
+KCl
ʵ�鷽��:һ�����ļױ���KMnO4��Һ��100 �淴Ӧһ��ʱ���ֹͣ��Ӧ,���������̷����������ͻ���δ��Ӧ�ļױ���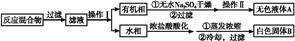
��֪:�����������122,�۵�122.4 ��,��25 ���95 ��ʱ�ܽ�ȷֱ�Ϊ0.3 g��6.9 g;���������л���һ�㶼�й̶��۵㡣
(1)��������������,����������������
(2)��ɫҺ��A����������,���Լ���A���Լ�����������,�������� ��
(3)�ⶨ��ɫ����B���۵�,��������115 �濪ʼ�ۻ�,�ﵽ130 ��ʱ�����������ۡ���ͬѧ�Ʋ��ɫ����B�DZ�������KCl�Ļ����,��������·��������ᴿ�ͼ���,ʵ���������Ʋ���ȷ������ɱ������ݡ�
| ��� | ʵ�鷽�� | ʵ������ | ���� |
| �� | ����ɫ����B����ˮ��,�����ܽ�,������ | �õ���ɫ�������ɫ��Һ | |
| �� | ȡ������Һ���Թ���,�������� | ���ɰ�ɫ���� | ��Һ��Cl- |
| �� | �����ɫ����,������ | ������ | ��ɫ���� �DZ����� |
(4)���Ȳⶨ:��ȡ1.220 g��Ʒ,���100 mL�״���Һ,��ȡ25.00 mL��Һ,�ζ�,����KOH�����ʵ���Ϊ2.40��10-3 mol����Ʒ�б��������������ļ������ʽΪ��������,������Ϊ��������(������λ��Ч����)��
TiO2�����Ʊ��������ѻ������ԭ�ϣ�����һ����������İ�ɫ���ϡ�
(1)ʵ�������÷�ӦTiO2(s)��CCl4(g)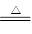 TiCl4(g)��CO2(g)������ˮ�����������Ʊ�TiCl4��ʵ��װ��ʾ��ͼ���£�
TiCl4(g)��CO2(g)������ˮ�����������Ʊ�TiCl4��ʵ��װ��ʾ��ͼ���£�
�й������������±���
| ���� | �۵�/�� | �е�/�� | ���� |
| CCl4 | ��23 | 76 | ��TiCl4���� |
| TiCl4 | ��25 | 136 | ����ʪ������������ |
(2)��ҵ����������(FeTiO3)(��Fe2O3��SiO2������)�Ʊ�TiO2���йط�Ӧ������
���ܡ�FeTiO3(s)��2H2SO4(aq)=FeSO4(aq)��TiOSO4(aq)��2H2O(l)
ˮ�⡡TiOSO4(aq)��2H2O(l)
 H2TiO3(s)��H2SO4(aq)
H2TiO3(s)��H2SO4(aq)��Ҫ�����������£�

���Լ�AΪ________����Һ������ȴ��70 �����ң����¶ȹ��ᵼ�²�ƷTiO2���ʽ��ͣ�ԭ����_________________________________________________
_______________________��
��ȡ������ϴ���H2TiO3���������Ტ���μ�KSCN��Һ�������������ټ�H2O2�������ɫ��˵��H2TiO3�д��ڵ�����������________������H2TiO3��ʹ��ˮ���ϴ�ӣ����պ��õ�TiO2Ҳ�ᷢ�ƣ����Ƶ�������____________________(�ѧʽ)��
1�������������������ϣ�Ҳ�ɺϳ��������ϡ�ʵ�����Ʊ�1�����������Ĺ������£� 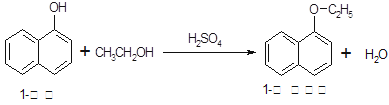
��֪����������ʵ���������
| ���� | ��Է� ������ | ״̬ | �۵�(��) | �е�(��) | �ܽ�� | |
| ˮ | �Ҵ� | |||||
| 1������ | 144 | ��ɫ���ɫ���νᾧ���ĩ | 96�� | 278�� | ����ˮ | �������Ҵ� |
| 1���������� | 172 | ��ɫҺ�� | 5��5�� | 267�� | ������ˮ | �������Ҵ� |
| �Ҵ� | 46 | ��ɫҺ�� | -114��1�� | 78��5�� | ����Ȼ��� | |
��1�����ӵ������뱽�����ƣ������ŵı�����ζ�������ڿ������ױ�����Ϊ�ۺ�ɫ��
��ش��������⣺
��1����72g1����������100mL��ˮ�Ҵ��У�����5mLŨ�����ϡ������Һ������ͼ��ʾ�������м��ȳ�ַ�Ӧ��ʵ����ʹ�ù����Ҵ���ԭ���� ��

��2����Ӧ����������ƿ�е�Һ�嵹����ˮ�У��������õ��л��㡣Ϊ�ᴿ�����������IJ�������������ˮϴ����Һ������10%��NaOH��Һ��ϴ����Һ��������ˮ�Ȼ��Ƹ��ﲢ���ˡ���ȷ��˳���� (�����)��
A���ۢڢܢ� B���٢ڢۢ� C���ڢ٢ۢ�
��3��ʵ����1�����������IJ����뷴Ӧʱ�䡢�¶ȵı仯��ͼ��ʾ��ʱ���ӳ����¶����ߣ�1�����������IJ����½���ԭ������� ��
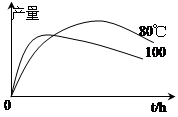
��4���ᴿ�IJ�Ʒ���ⶨΪ43g����ʵ����1�����������IJ���Ϊ ��