��Ŀ����
ijУ��ѧС���ͬѧ��չ��һϵ�еĻ�ѧʵ����
�������ʵ�鲢�����������:
(1)��ͬѧ��ͼ1��ʾװ��,����п�����ᷴӦ����:��2 gп��������ƿ��,ͨ����Һ©������1 mol��L-1ϡ����40 mL,�ռ�10 mL����,ͨ����¼�������������õ���Ӧ����Ϊx mol��(L��min)-1��ʵ�鿪ʼʱ����װ�������Եķ�������������������������������
(2)��ͬѧ��ͼ2װ�òⶨNa2CO3��NaCl�Ĺ���������Na2CO3����������:
�ټס������Թܸ�����������,�������Ӷ�Ӧ�ӿں�,����ʢϡ������Թ�,������Ӧ,�ų�����,����������ϡ����Ӧ�ֱ�����������������������(���������);
��G�ܿ����û�ѧʵ�������һ�ֳ������������,����������������;
�������ס��ҽӿڵ����ӷ�ʽ����:A��������,B����������,C����������(��д���ӿڵı��);
��Ϊ��߲�����ȷ��,�ռ��������,��װ�ö���ǰӦ���еIJ�����������������������������������������������������
(3)��ͬѧ���ͬѧʵ��Ŀ����ͬ:��ͼ3װ�òⶨ���ɵ�CO2������,����װ�ô�������ȱ��,�Ӷ�����ʵ�����,�����������ʹ�ⶨ�������ƫ�����Ҫԭ��������������������������������
(1)��Ӧʱ�䡡�ر�A������,��ע������������һ������,һ��ʱ����ɿ�����,�������ܻص�ԭλ,֤����©��,����©��
(2)�ټס��ҡ��ڼ�ʽ�ζ��ܡ���D��E��F���������ƶ��ζ���,ʹ��������Һ����ƽ��ƫ��
(3)CO2�����л���ˮ����������е�CO2��ˮ��������������
����

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д���������ƵĴ��ȿ��õζ������вⶨ��ԭ���ǣ�2S2O32��+ I2 �� S4O62��+ 2I-
38.����100 mL0.0500 mol/L I2��Һ������Ҫ�������� ��ѡ���ţ���
a��100 mL����ƿ b����Ͳ c���ձ� d��������
�ζ��ܱ���ʹ���¶ȣ�20oC; �ζ��ܵ���С�̶�Ϊ mL��
39.ȡ2 .500g�����ʵ�Na2S2O3��5H2O�������50mL��Һ��ÿ��ȡ10.00mL������ƿ���2�ε���Ϊָʾ��������0.0500 mol/L I2��Һ�ζ���ʵ����������(��3�γ�����Ϊ 0.00���յ������ͼ; ���ʲ��μӷ�Ӧ)��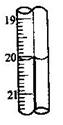
| ��� | 1 | 2 | 3 |
| ����I2��Һ�����/mL | 19.98 | 20.02 | |
Na2S2O3?5H2O(ʽ��248)�����������ǣ�����4λС���� ��
40.�����ʵ����ƫ�͵IJ����� ��ѡ���ţ���
a. �ζ�ʱ�ζ����е�Һ�������ƿ��
b. ��ƿ������ˮϴ��������װ�������Һ
c. δ�ñ�Һ��ϴ�ζ���
d. ��ʱ��Һ������ƿ��
����ͭ�����ڲ�ͬ�¶��¿�ʧȥ���ֻ�ȫ���ᾧˮ�����ֽ�������ijѧ���ڲ�ͬ�¶��¸�8.000 g����ͭ������ȣ��¶������ߣ���ʵ������¼���£�
| ʵ����� | �¶ȣ��棩 | ��ȴ��ʣ������������g�� |
| 1 | 102 | 6.848 |
| 2 | 113 | 5.696 |
| 3 | 258 | 5.120 |
| 4 | 570 | 2.560 |
��1����1��ʵ�����������ʧȥ�ᾧˮ��������_____ g��
��2��ͨ������ȷ���ڶ���ʵ���������ʵĻ�ѧʽΪ______________��
��3�����Ĵ�ʵ���ʣ�����Ϊһ�ֺ�ɫ��ĩ������Ϊ����������ú�ɫ��ĩ�ܽ���ϡ�����У�������Ҫ0.50mol/L��ϡ������ٺ�������д��������̣�
̼��������ҹ���Ҫ�ĵ���Ʒ��֮һ���������������������ӷ���ʧ��Ϊ�˼�����������ȷ�����ʩ����������ⶨ�京������
��ijѧ�������һ���Բⶨ������̼������Ӳⶨ�������ķ���������Ʒ����Բ����ƿ�У�

��1����ѡ���Ҫ��װ�ã���������������˳��Ϊ ��
��2����Һ©���е�Һ�����ʺϵ��� ��
| A��ϡ���� | B��ϡ���� | C��Ũ���� | D���������� |
����������гɷ��ǣ�NH4��2SO4��������ü�ȩ���ⶨ����������ȩ���ǻ��ڼ�ȩ��һ������������ã������൱�����ᣬ��ӦΪ2��NH4��2SO4+6HCHO����CH2��6N4 +2H2SO4 + 6H2O,���ɵ��������������Ʊ���Һ�ζ����Ӷ��ⶨ���ĺ������������£�
��1���ò�������ȡ���壨NH4��2SO4��Ʒ0��6g���ձ��У�����Լ30mL����ˮ�ܽ⣬�������100mL��Һ���� �����ʽ����ʽ�����ζ���ȷȡ��20��00mL����Һ����ƿ�У�����18%���Լ�ȩ��Һ5mL������5min����1~2�� ָʾ������֪�ζ��յ��pHԼΪ8��8������Ũ��Ϊ0��08mol/L�������Ʊ���Һ�ζ����������±���
| �ζ����� | �ζ�ǰ������mL�� | �ζ��������mL�� |
| 1 | 1��20 | 16��21 |
| 2 | 3��00 | 18��90 |
| 3 | 4��50 | 19��49 |
��ζ��յ�ʱ������Ϊ ���ɴ˿ɼ��������Ʒ�еĵ�����������Ϊ ��
��2���ڵζ�ʵ��������ֵζ��õļ�ʽ�ζ��ܲ��������ڳ��������ݣ��ζ���ʼʱ�����ݣ����ʵ��ⶨ�ĺ�������ʵ��ֵ ���ƫ��ƫС������Ӱ�족����
������ⶨ̼������еĺ�����ʱ��ʹ�ü�ȩ���Ƿ���� ����ǡ����������� ��
�Ȼ��Ƴ����ڵ�·�ڱ��������������������ˮ�������������ʡ�ʵ�����ù�ҵ����ʯ����������Al2O3��Fe2O3�����ʣ��Ʊ��Ȼ��Ƶ���Ҫ�������£�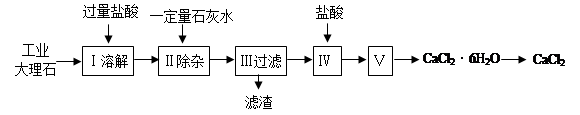 ���������գ�
���������գ�
��1������ʹ�õ���������ʵ���Ũ��ԼΪ6.0mol/L������36.5%�����ᣨ�ܶ�Ϊ1.2g/mL������6.0mol/L������100mL������IJ��������в���������Ͳ����ͷ�ιܡ� ����Ҫ��ȡ36.5%������ mL�����ƹ����У���������������ȷ�����в���������Ũ��ƫС���� ��
| A������ҡ�Ⱥ���Һ����ڿ̶��� |
| B������ʱ��������ƿ�Ŀ̶��� |
| C������Һת������ƿ��û��ϴ���ձ��Ͳ���������ת�붨�ݲ��� |
| D�������ˮ�����˿̶��ߣ�ȡ������ˮʹҺ��ǡ�õ��̶��� |
��3����������ʯ��ˮ������Ŀ���Ƿ�ֹ�������ӷ���ʽ��ʾ���� ��
��4����������Ҫ�ɷ��У�д��ѧʽ�� ��
��5������������IJ��������� �� ��
��6���Ƶõ��Ȼ��ƿ���ͨ�����²���ⶨ�䴿�ȣ�
�ٳ�ȡ5.5g��ˮ�Ȼ�����Ʒ�����ܽ⣻�۵�������Na2CO3��Һ����ַ�Ӧ����ˣ��� �� ���ݳ��������õ�4.3g̼��ƣ����Ȼ��Ƶ���������Ϊ ����ʵ������淶���ⶨ���ƫ�ͣ���ԭ���� ��
�����Ƿ�����������FeS2���������ַ������������IJ�������ͼ���£�
��ش��������⣺
(1)����ͼ�в����١��ڡ��۷ֱ�ָ���Ǣ�_________����__________����________��
�����ܡ����õ�����Ҫ�����ǣ���__________����__________(ÿ����1-2������)��
(2)�ж���Һ��SO42-�����ѳ�����ȫ�ķ�����_________________________________��
(3)ijͬѧ�÷�����ⶨ������FeԪ�صĺ�����ȷ��ȡһ�����Ŀ�ʯ�������������ܽ⡢Ԥ������
| A���ô��п̶ȵ��ձ����Ƴ�100 mL������Һ�� | B������Ͳ��ȡ25.00 mL������Һ�� | C����������ƿ�С� | D��������ˮϴ�ӵζ��ܺ�װ��KMnO4����Һ���øñ���Һ�ζ�����������(E)����Һ��ɵ��Ϻ�ɫʱ��ֹͣ�ζ�����30���ڲ���ɫ��(F)��ȡ������ζ��������ĵ�KMnO4����Һ��������������е�FeԪ�غ�������ָ����ʵ������д����������ı��________________________�� |
(5)��ȡ��ʯ����1.60 g, ��������������Ƶ�BaSO4������Ϊ4.66 g�������ʯ�е���Ԫ��ȫ��������FeS2����ÿ�ʯ��FeS2������������___________��
�Ͼ���Ļ������ü������ڽ�Լ��Դ���������ڱ���������ij�о�С��ͬѧ�ԷϾ�п�̸ɵ��Ϊԭ�ϣ����Ͼɵ�غ�п����ת����ZnSO4��7H2O�����̲���ת���ɴ��Ƚϸߵ�MnO2����NH4Cl��ҺӦ���ڻ��������У�ʵ���������£�
��1�������������õļ�������Ӧѡ ��ѡ�����������������
��2������ҺA�����ĵ�һ���Ǽ��백ˮ����pHΪ9��ʹ���е�Fe3+��Zn2+��������д����ˮ��Fe3+��Ӧ�����ӷ���ʽ ��
��3����������Ϊ�˳�ȥ��Һ�е�Zn2+����֪25��ʱ��
| NH3��H2O��Kb | Zn2+��ȫ������pH | Zn(OH)2���ڼ��pH |
| 1.8��10��5 | 8.9 | ��11 |
���ϱ����ݷ���Ӧ������ҺpH���Ϊ ������ţ���
a��9 b��10 c��11
��4�� MnO2����������Ҫ���裺
����1����3%H2O2��6.0mol/L��H2SO4�Ļ��Һ����MnO2�ܽ⣬���ȳ�ȥ����H2O2����MnSO4��Һ��������Fe3+������Ӧ����MnSO4�����ӷ���ʽΪ ��
����2����ȴ�����£��μ�10%��ˮ����pHΪ6��ʹFe3+������ȫ���ټӻ���̿���裬���ˡ��ӻ���̿�������� ��
����3������Һ�еμ�0.5mol/L��Na2CO3��Һ������pH��7���˳�������ϴ�ӡ�����������ں�ɫ������MnO2�����չ����з�Ӧ�Ļ�ѧ����ʽΪ ��
��5�� ������֪����MnO2���ܽ�������������������ݣ�Ȼ����ȡMnCO3���塣
���������������Һ��Ũ�Ⱦ�Ϊ5mol/L�������Ⱥ���ѽ���ʱ���£������¶ȶ�MnCO3���ʵ�Ӱ����ͼ4����ͼ�������������ѽ����¶ȶ��� �����ң�
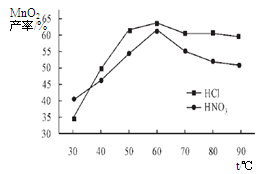
��������¶ȡ���ѽ���ʱ����������£����Ũ�ȶ�MnCO3���ʵ�Ӱ����ͼ5����ͼ������������Ũ��Ӧѡ�� mol/L���ҡ�

