��Ŀ����
��t��ʱ��a g NH3��ȫ����ˮ���õ�V mL��Һ���������Һ���ܶ�Ϊd g/cm3����������Ϊ�������к�NH4�������ʵ���Ϊb mol������������ȷ����(����)
| A�����ʵ���������Ϊ������100% |
| B����ˮ�����ʵ���Ũ��Ϊ1000a/(35V)mol��L��1 |
| C����Һ��c(OH��)��1000b/V mol��L��1��c(H��) |
| D��������Һ���ټ���V mLˮ��������Һ��������������0.5�� |
C
A����ˮ��Һ����Ϊ����������Һ���ܶ�Ϊ�� g?cm-3�����ΪVmL��������Һ����Ϊ��Vg�����ʰ���������Ϊag�����ʵ���������Ϊag /(��Vg) ��100%����A����
B��ˮ���ܶȱȰ�ˮ���ܶȴ��������İ�ˮ��ˮ��ˮ�������������Ϻ���Һ����������ԭ��ˮ��2������Һ�а�����������ͬ����������������Һ���ʵ���������С��0.5w��
��B����
C����ҺOH-����Դ��һˮ�ϰ���ˮ�ĵ��룬NH4+��Ũ��Ϊbmol /(V��10-3L) ="1000b" /V (mol/L)��һˮ�ϰ�����NH3?H2O?NH4++OH-��һˮ�ϰ��������OH-Ϊ1000b /V (mol/L)��������Һ��OH-Ũ�ȴ���1000b /V (mol/L)����C����
D��a g NH3�����ʵ���Ϊag /(17g/mol) ="a" /17 (mol)����Һ���ΪVmL��������Һ�����ʵ���Ũ��Ϊ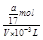 =
=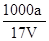 mol/L����D��ȷ��
mol/L����D��ȷ��
��ѡ��D��
�������������ʵ���Ũ�ȡ����������ļ��������ϵ�ȣ��Ѷ��еȣ���������ǽ���Ĺؼ���
B��ˮ���ܶȱȰ�ˮ���ܶȴ��������İ�ˮ��ˮ��ˮ�������������Ϻ���Һ����������ԭ��ˮ��2������Һ�а�����������ͬ����������������Һ���ʵ���������С��0.5w��
��B����
C����ҺOH-����Դ��һˮ�ϰ���ˮ�ĵ��룬NH4+��Ũ��Ϊbmol /(V��10-3L) ="1000b" /V (mol/L)��һˮ�ϰ�����NH3?H2O?NH4++OH-��һˮ�ϰ��������OH-Ϊ1000b /V (mol/L)��������Һ��OH-Ũ�ȴ���1000b /V (mol/L)����C����
D��a g NH3�����ʵ���Ϊag /(17g/mol) ="a" /17 (mol)����Һ���ΪVmL��������Һ�����ʵ���Ũ��Ϊ
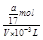 =
=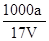 mol/L����D��ȷ��
mol/L����D��ȷ����ѡ��D��
�������������ʵ���Ũ�ȡ����������ļ��������ϵ�ȣ��Ѷ��еȣ���������ǽ���Ĺؼ���

��ϰ��ϵ�д�
�����Ŀ
 �İ�ˮ��
�İ�ˮ��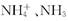 ��
��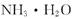 ������Ϊ
������Ϊ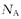
 ϡ����
ϡ����