��Ŀ����
������������Ҫ�Ĺ�ҵԭ�Ϻ��ܼ�����������ͼװ����ȡ���������Ĵֲ�Ʒ���ٲⶨ���������ĺ�����

(1)д����Ӧ����ʽ________(��Ӧ)
(2)ʵ����һ���ñ���̼������Һ���շ�Ӧ��������������Թܣ��ɿ�����������________�������Թܣ��ɿ�����������________��������ȴ�����˰�ɫ���壬�þ�����________��
(3)��������������������װ�ã�Ϊ����߲��ʣ������һ���Ľ������
________��
(4)Ϊ�ⶨ���������ĺ������������²�����
(��)ȷ����20.0 g����������Ʒ����ƿ�У���0.50 mol/L��NaOH�ζ�(��̪��ʾ��)���յ�ʱ����NaOH��Һ�����Ϊ40.0 mL
(��)��ȡ20.0 g��������ֲ�Ʒ��250 mL��ƿ�У�����100 mL��2.1 mol/L��NaOH��Һ�����ɫ�Ⱥ�װ�������ܣ���ˮԡ�ϼ��Ȼ���Լ1Сʱ��װ����ͼ��ʾ������ȴ����0.50 mol/LHC1�ζ�������NaOH���յ�ʱ������������Ϊ20.0 mL��
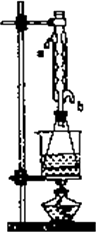
�ش𣺢�ʵ��(��)�дﵽ�ζ��յ�ʱ��������________��
��ʵ��(��)����ˮ����ˮ���ܵ�________(��a��b)�ܿ�ͨ�룮
������ʵ��(��)��(��)���������ݼ���ֲ�����������������������Ϊ________��
������
����(1)CH3COOH��C2H5OH![]() CH3COOC2H5��H2O���ữ��Ӧ
CH3COOC2H5��H2O���ữ��Ӧ
����(2)��ɫҺ���Ϊ���㱣�ֲַ㣬���������ݳ���NaHCO3
����(3)ˮԡ���Ȼ�����������
����(4)����Һ����ɫǡ�ñ�Ϊdz��ɫ
������b
������79.2��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�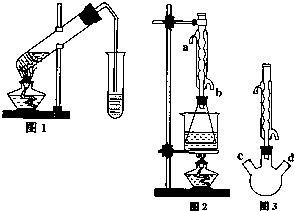 ������������Ҫ�Ĺ�ҵԭ�Ϻ��ܼ���ͨ�����������Ҵ������ᣮijѧϰС������ͼ1װ����ȡ���������ֲ�Ʒ���ٷ������������ĺ�����
������������Ҫ�Ĺ�ҵԭ�Ϻ��ܼ���ͨ�����������Ҵ������ᣮijѧϰС������ͼ1װ����ȡ���������ֲ�Ʒ���ٷ������������ĺ�����| �������� | �Ҵ� | ���� | |
| �е� | 77.1�� | 78.5�� | 117.9�� |
��I��ȷ����20.0g����������Ʒ����ƿ�У���0.50mol?L-1NaOH�ζ�����̪��ָʾ�������յ�ʱ����NaOH��Һ�����Ϊ40.0mL
��II����ȡ20.0g���������ֲ�Ʒ��250mL��ƿ�У�����100mL 2.1mol?L-1NaOH��Һ��Ͼ��Ⱥ�װ�������䣬��ˮԡ�ϼ��Ȼ���Լ1Сʱ��װ����ͼ2��ʾ������ȴ����0.50mol?L-1HCl�ζ�������NaOH���յ�ʱ������������Ϊ20.0mL��
�ش��������⣺
��1��ʵ�飨II������ˮ����ˮ���ܵ�
��2������ʵ�飨I������II�����������ݼ���ֲ�����������������������Ϊ
��3��ʵ�������ͬѧ�ǶԴֲ�Ʒ�����������ĺ������߽������ۣ�
��������Ϊ��ʵ�飨II�������������齫ͼ2�е���ƿ��Ϊ����ƿ��װ����ͼ3��������ƿ��c��d��װ����ص�����������ǡ���IJ�����������߲ⶨ�ľ�ȷ�ȣ�����Ϊ������ƿ��c��d��װ����ص�����������ǣ�
A��װ���¶ȼƣ��ϸ���Ʒ�Ӧ�¶�
B��ʵ���о�����ƿ�ڣ��ò������н���
C���ڷ�Ӧ���ڣ������Ȱ�װ�ķ�Һ©������һ������NaOH��Һ
�ڻ���ͬѧ��Ϊ�Ľ�������������ȡװ�ã�ͼ1��������߲��ʣ�������һ���Ľ�����
 ������Ҫ���л��ϳ��м��壬�㷺Ӧ�����ܼ������ܼ������ϡ�ճ�ϼ���ӡˢ����֯�ȹ�ҵ������������ʵ���Һ�ҵ�Ʒ����������·�Ӧ��CH3COOH+C2H5OH
������Ҫ���л��ϳ��м��壬�㷺Ӧ�����ܼ������ܼ������ϡ�ճ�ϼ���ӡˢ����֯�ȹ�ҵ������������ʵ���Һ�ҵ�Ʒ����������·�Ӧ��CH3COOH+C2H5OH 


 (08�Ϻ��ɽ���ģ��)������������Ҫ�Ĺ�ҵԭ�Ϻ��ܼ�����������ͼ
(08�Ϻ��ɽ���ģ��)������������Ҫ�Ĺ�ҵԭ�Ϻ��ܼ�����������ͼ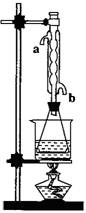 �Ƕ�������������������װ�ã�Ϊ����߲��ʣ������
�Ƕ�������������������װ�ã�Ϊ����߲��ʣ������