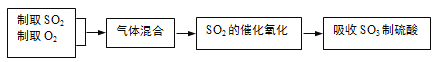
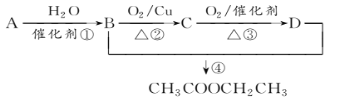
��Ŀ����
����Ŀ���±���Ԫ�����ڱ���һ���֣���ش��й����⣺
���� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A |
2 | �� | �� | �� | �� | |||
3 | �� | �� | �� | �� | |||
4 | �� | �� |
��1����2���ڵ�8��Ԫ���У���һ�����ܽ��ڢ�Ԫ�غ͢�Ԫ�ؼ��Ԫ����__�֡�
��2���������γ��������������Ԫ����___����Ԫ������![]() ��д����Ԫ�صĵ����������������ˮ���ﷴӦ�����ӷ���ʽ___��
��д����Ԫ�صĵ����������������ˮ���ﷴӦ�����ӷ���ʽ___��
��3���ڡ��ݡ��ߡ�������Ԫ�ص�����������ˮ�����У������Լ���������ǿ��˳������Ϊ���û�ѧʽ��ʾ��___��
��4����Ԫ�����Ԫ������������֮����___��
��5����д���۵������⻯����������Ļ�ѧ����ʽ___��
��6����д����Ԫ�ص�����������ˮ�������Ԫ�ص�����������ˮ���ﷴӦ�����ӷ���ʽ___��
���𰸡�3 �� 2Al+OH-+2H2O=2AlO2-+3H2�� KOH��Mg(OH)2��H2CO3��H2SO4 26 4NH3+5O2 4NO+6H2O Al(OH)3+3H+=Al3++3H2O
4NO+6H2O Al(OH)3+3H+=Al3++3H2O
��������
(1)��ΪAl����N��ͬһ����Ԫ���У�Ԫ�صĵ�һ����������ԭ�����������������������ƣ�Mgԭ�������Ϊȫ��״̬�����Ե�һ�����ܴ���Al��Nԭ�������Ϊ����״̬�����Ե�һ�����ܴ���O�����Ե�һ�����ܽ���Al��N��Ԫ����Mg��C��O����3�֣�
(2)��Ԫ�����γ��������������ΪK�������������ˮ����ΪKOH��Al������ǿ�Ӧ����������ƫ����Σ��������ӷ���ʽΪ2Al+OH-+2H2O=2AlO2-+3H2����
(3)�ڡ��ݡ��ߡ���ֱ�ΪC��Mg��S��K��������Խǿ������������ˮ����ļ���Խǿ��������K>Mg>C>S�����Լ��Լ���������ǿ��˳������ΪKOH��Mg(OH)2��H2CO3��H2SO4��
(4)��ΪFΪ9��Ԫ����ԭ�Ӻ�9�����ӣ���ΪBr��Ϊ35��Ԫ����ԭ�Ӻ�35�����ӣ�������֮��Ϊ26��
(5)��ΪN���������⻯��ΪNH3������������NO��ˮ������ʽΪ4NH3+5O2 4NO+6H2O��
4NO+6H2O��
(6)��ΪAl������������ˮ����ΪAl(OH)3����ΪCl���������ΪΪHClO4����Ӧ�����ӷ���ʽΪ��Al(OH)3+3H+=Al3++3H2O��

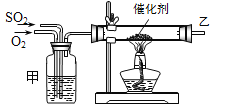
����Ŀ����ҵ����������(��Ҫ�ɷ�ΪAl2O3����������SiO2��FeO��xFe2O3������)��ȡ����һ�ֹ�������ʾ��ͼ���£�
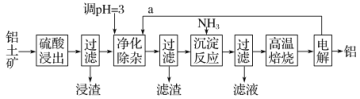
��֪�������������������pH���±���
������ | Fe(OH)3 | Al(OH)3 | Mg(OH)2 |
pH | 3.2 | 5.2 | 12.4 |
(1)Ϊ��߽������ʣ����ʵ���������Ũ���⣬���ɲ�ȡ�Ĵ�ʩ��_______________
(2)��������Ҫ�ɷ�Ϊ____________________
(3)�������Ӳ�����Ϊ��������һ����ͨ������a����Ŀ����_____________________���ڶ����ǿ�����ҺpH��ֻʹFe3+ת��ΪFe(OH)3�����������������ɵij����л�������Һ�е��������ʣ���Һ�е��������ʱ���ͬ������ԭ����_____________________
(4)д��������Ӧ�����з�����Ӧ�����ӷ���ʽ��________________________��
(5)ʵ���Ҹ��±���ʱ������ʢ�Ź��������������_______________��
(6)��AlCl3��MgCl2�Ļ��Һ�еμ�NaOH��Һ�����ɳ������������NaOH��Һ�������ϵ��ͼ��ʾ����ԭ��Һ��AlCl3��MgCl2�����ʵ���֮��Ϊ__________����д������NaOH��Һ�����Ϊ50-60mLʱ�Ļ�ѧ��Ӧ����ʽ__________________________��