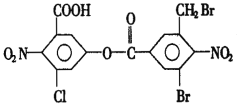
��Ŀ����
����Ŀ��һ�ָ�Ч��ˮ�����������ۺ��Ȼ�������Ļ�ѧʽΪ[Al2(OH)nCl6��n��xH2O]m������ͨ������AlCl3��Һ��pH���ٽ���ˮ����ᾧ���������Ʊ�ԭ����Ҫ�����ӹ���ҵ�ķ����������ң�����Ҫ��Al2O3��Al������SiO2�����ʡ��ۺ��Ȼ��������Ĺ����������£�
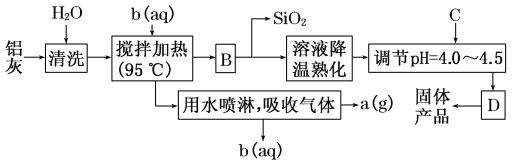
��1��������Ȳ��������з�����Ӧ�����ӷ���ʽΪ_______________________________________��
��2������������ʵ�����B��D�����ƾ�Ϊ________��
��3����Ӧ�и���Ʒa��________���Լ�b��________��(�û�ѧʽ��ʾ)
��4�����������п�ѭ��ʹ�õ�������________(�û�ѧʽ��ʾ)��
��5������pH��4.0��4.5��Ŀ����______________________________________________��
��6��Ϊ�õ��ϴ����ľ��壬����������C���ʿ�ѡ��________��
A����ˮ�� B��NaAlO2�� C��NaOH D��Al2O3�� E��Al
���𰸡�Al2O3��6H��===2Al3����3H2O��2Al��6H��===2Al3����3H2�� ���� H2 HCl HCl �ٽ�AlCl3ˮ�⣬ʹ�������� DE
��������
������Ҫ��Al2O3��Al������SiO2�ȣ���ˮ��ϴ���������������ʣ���ΪB�������������裬�����Ƴ�b��ҺΪ������Һ�����������Ὣ��������������ת��Ϊ�Ȼ�����Һ��ͬʱ������������Ϊ�Ȼ����ӷ������������л����Ȼ��⣬��ˮ���ܣ��õ�����aΪ������ͬʱ��������b��BΪ�Ȼ�����Һ�л��в����ԵĶ������裬ͨ�����˷�����������裬�Ȼ�����Һ��Ũ�������pH�����á����˵õ��ۺ��Ȼ������塣
��1���ɷ�����֪bΪ������Һ��������������ת��Ϊ�Ȼ�����Һ�����Խ�����Ȳ��������з�����Ӧ�����ӷ���ʽΪ��Al2O3��6H��=2Al3����3H2O��2Al��6H��=2Al3����3H2�����ʴ�Ϊ��Al2O3��6H��=2Al3����3H2O��2Al��6H��=2Al3����3H2����
��2���ɷ�����֪������������ʵ�����B��D�����ƾ�Ϊ���ˣ��ʴ�Ϊ�����ˣ�
��3���ɷ�����֪��aΪ������bΪ���ᣬ�ʴ�Ϊ��H2��HCl��
��4��������ȹ�������Ҫ�������ᣬ������������пɻ��ջӷ�����HCl������HCl����ѭ��ʹ�ã��ʴ�Ϊ��HCl��
��5���Ȼ���ˮ������ԣ����Ե���pH��Ŀ����Ϊ��ʹ�Ȼ���ˮ�����ɲ�Ʒ���ʴ�Ϊ���ٽ�AlCl3ˮ�⣬ʹ����������
��6��Ϊ�õ��ϴ����ľ��壬����pH���Լ��������������ʣ��������İ�ˮ��NaAlO2��NaOH�����׳�ȥ�������������ʣ� Al2O3��Al��Ϊ������ˮ������ʱ��ͨ�����˳�ȥ��������Al2O3��Al����pH�ܵõ��ϴ����ľ��壬������������ΪDE��

����Ŀ������ʵ��������ʵ���������ƥ�����
ʵ����� | ʵ������ | |
A | ��ʢ�и������������Һ���Թ���ͨ����������ϩ���� | ��Һ����ɫ����ȥ�����ú���Һ�ֲ� |
B | ��þ����ȼ��Ѹ�����뼯��CO2�ļ���ƿ | ����ƿ�в���Ũ�̲��к�ɫ�������� |
C | ��ʢ�б��������������Һ���Թ��еμ�ϡ���� | �д̼�����ζ�����������Һ����� |
D | ��ʢ��FeCl3��Һ���Թ��мӹ������ۣ�������1��KSCN��Һ | ��ɫ����ʧ����KSCN����Һ��ɫ���� |
A. AB. BC. CD. D
����Ŀ����������ʵ���ó�����Ӧ������ȷ����
ʵ����ʵ | ���� | |
A | ����ͬ�¶��£���1 mL0.2 mol/LNaOH��Һ�е���2��0.1 mol/LMgCl2��Һ��������ɫ�������ٵμ�2��0.1 mol/LFeCl3��Һ�������ɺ��ɫ���� | �ܽ�ȣ�Mg(OH)2>Fe(OH)3 |
B | ij������ʹʪ�����ɫʯ����ֽ��� | ������ˮ��Һһ���Լ��� |
C | ͬ��ͬѹ�£������pH=3��HA��HB������ֱ���������п��Ӧ����ˮ���ռ����壬HA�ų����������ҷ�Ӧ���ʿ� | HB�����Ա�HAǿ |
D | SiO2����������ᷴӦ������Ӧ | SiO2������������ |
A.AB.BC.CD.D