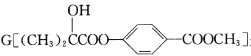
��Ŀ����
����Ŀ����1 mol N2�� 3 mol H2��������ɱ�ĺ����ܱ�������,��380 ���·�����Ӧ��N2(g)+3H2(g)![]() 2NH3(g)��ƽ��ʱ,��ϵ�а����������
2NH3(g)��ƽ��ʱ,��ϵ�а����������![]() (NH3)��ѹǿ�仯��������±�:
(NH3)��ѹǿ�仯��������±�:
ѹǿ/MPa | 10 | 20 | 30 | 40 |
| 0.30 | 0.45 | 0.54 | 0.60 |
����˵����ȷ����(����)
A.10 MPaʱ,H2��ת����Ϊ75%
B.20 MPaʱ,NH3�����ʵ���Ũ����10 MPaʱ��1.5��
C.40 MPaʱ,�����������ΪV L,��ƽ�ⳣ��K=![]()
D.30 MPaʱ,���������г����������,��ƽ��������Ӧ�����ƶ�
���𰸡�C
��������
A.10 MPaʱ��
N2(g)+3H2(g)![]() 2NH3(g)
2NH3(g)
��ʼ�� 1 mol 3 mol 0
�仯�� x 3x 2x
ƽ���� 1-x 3-3x 2x
��![]()
x=![]() ��H2��ת����Ϊ
��H2��ת����Ϊ![]() ��A����
��A����
B. 20 MPaʱ��
N2(g)+3H2(g)![]() 2NH3(g)
2NH3(g)
��ʼ�� 1 mol 3 mol 0
�仯�� y 3y 2y
ƽ���� 1-y 3-3y 2y
��![]()
y=![]() ��NH3�����ʵ���Ũ����10 MPaʱ��
��NH3�����ʵ���Ũ����10 MPaʱ�� ��1.5����B����
��1.5����B����
C. 40 MPaʱ��
N2(g)+3H2(g)![]() 2NH3(g)
2NH3(g)
��ʼ�� 1 mol 3 mol 0
�仯�� z 3z 2z
ƽ���� 1-z 3-3z 2z
��![]()
z=![]() ��K=
��K=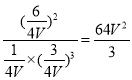 ��C��ȷ��
��C��ȷ��
D. 30 MPaʱ�����������г���������壬�������������Ӧ����������Ũ��ͬ�ȳ̶ȼ�С����ƽ�����淴Ӧ�����ƶ���D����
��ѡC��

 ��У����ϵ�д�
��У����ϵ�д�����Ŀ����ҵ�ϳɰ���ӦΪ��N2��g��+3H2��g��![]() 2NH3��g���������о����£�
2NH3��g���������о����£�
��1����֪H��H����Ϊ436kJ��mol��1��N��H����Ϊ391kJ��mol��1��N��N���ļ�����946kJ��mol��1����������Ӧ����H��_________________��
��2��������Ӧ��ƽ�ⳣ��K�ı���ʽΪ____________������Ӧ����ʽ��дΪNH3��g��![]() N2��g��+
N2��g��+![]() H2��g������ƽ�ⳣ��K1��____________________����K��ʾ����
H2��g������ƽ�ⳣ��K1��____________________����K��ʾ����
��3����773Kʱ���ֱ�2mol N2��6mol H2����һ���̶��ݻ�Ϊ1L���ܱ������У����ŷ�Ӧ�Ľ��У�����������n��H2����n��NH3���뷴Ӧʱ��t�Ĺ�ϵ���±���
t/min | 0 | 5 | 10 | 15 | 20 | 25 | 30 |
n��H2��/mol | 6.00 | 4.50 | 3.60 | 3.30 | 3.03 | 3.00 | 3.00 |
n��NH3��/mol | 0 | 1.00 | m | 1.80 | 1.98 | 2.00 | 2.00 |
�ٱ�����m��_______________/span>��15��25min�ڣ�v��N2����_______________��
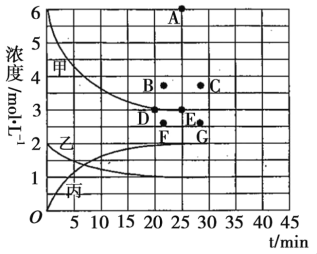
�ڸ��¶��£�����ͬ�ݻ�����һ������Ͷ���N2��H2��NH3Ũ�Ⱦ�Ϊ3mol��L��1����ʱv��_______v��������������������������������
���ɱ��е�ʵ�����ݼ���õ���Ũ�ȡ�ʱ�����Ĺ�ϵ����ͼ�е����߱�ʾ����ʾc��N2����t��������______________�������������������������������ڴ��¶��£�����ʼ����4mol N2��12mol H2����Ӧ�մﵽƽ��ʱ����ʾc��H2������������Ӧ�ĵ�Ϊ_________________��
��4��Marnellos��Stoukides���õ�ⷨ�ϳɰ���ʵ���˳�ѹ�ϳɺ͵����ĸ�ת���ʡ��÷�����SCY�մɽ�����������SCY�մɾ��и����ӵ����ԣ��������Ǵ���H�����������ĵ缫��ӦΪ____________________��