��Ŀ����
�������� ��Cu ��Һ̬SO2 �۴����� ��NaHCO3 ��Ba��OH��2��Һ
I�����ڷǵ���ʵ���______��������ţ�
��1��NaHCO3��һ��______���ǿ��������������ʣ�
��2��д��HCO3-ˮ������ӷ���ʽ��______��
��3�������£�0.1mol-L-1NaHCO3��Һ��pH����8������Һ��Na+��HCO3-��H2CO3��CO32-��OH -��������Ũ���ɴ�С��˳��Ϊ��______��
��Ba��OH��2��һ��ǿ����ʣ�����25�桢pH=13��Ba��OH��2��Һ��
��1����Ba��OH��2��Һ�����ʵ���Ũ��Ϊ______��
��2����Һ����ˮ�����c��OH -��=______��
��3����ijŨ��������Һ������ȣ�������֮�ȣ�1��9��Ϻ�������ҺpH=11����������Һ��������ڻ��ǰ����Һ������ͣ�����������Һ��pH=______��
����������һ��������ʣ� ij�¶��£���ͬpHֵ������ʹ�����Һ�ֱ��ˮϡ�ͣ�ƽ��pHֵ����Һ����仯����������ͼ��ʾ����ͼ______���I����Ϊ����ϡ�͵�ͼ��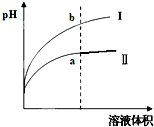
I�����ڷǵ���ʵ���______��������ţ�
��1��NaHCO3��һ��______���ǿ��������������ʣ�
��2��д��HCO3-ˮ������ӷ���ʽ��______��
��3�������£�0.1mol-L-1NaHCO3��Һ��pH����8������Һ��Na+��HCO3-��H2CO3��CO32-��OH -��������Ũ���ɴ�С��˳��Ϊ��______��
��Ba��OH��2��һ��ǿ����ʣ�����25�桢pH=13��Ba��OH��2��Һ��
��1����Ba��OH��2��Һ�����ʵ���Ũ��Ϊ______��
��2����Һ����ˮ�����c��OH -��=______��
��3����ijŨ��������Һ������ȣ�������֮�ȣ�1��9��Ϻ�������ҺpH=11����������Һ��������ڻ��ǰ����Һ������ͣ�����������Һ��pH=______��
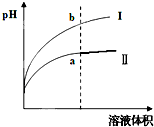
����������һ��������ʣ� ij�¶��£���ͬpHֵ������ʹ�����Һ�ֱ��ˮϡ�ͣ�ƽ��pHֵ����Һ����仯����������ͼ��ʾ����ͼ______���I����Ϊ����ϡ�͵�ͼ��

��Cu�ǽ��������ܵ��磬�����ʼ����ǵ����Ҳ���Ƿǵ���ʣ�
��Һ̬SO2�ǹ��ۻ����ﲻ�ܵ��磬���ǵ���ʣ����ڷǵ���ʣ�
�۴���������ˮ�ܵ��磬�ǵ���ʣ�
��NaHCO3�����ӻ�����ǵ���ʣ�
��Ba��OH��2��Һ�ǻ��������ǵ����Ҳ���Ƿǵ���ʣ�
��ѡ���ڣ�
��1��NaHCO3�����Σ���ˮ��Һ����ȫ���룬����ǿ����ʣ��ʴ�Ϊ��ǿ��
��2��HCO3-ˮ������̼�������������ӣ�ˮ�����ӷ���ʽΪHCO3-+H2O

H2CO3+OH-���ʴ�Ϊ��HCO3-+H2O

H2CO3+OH-��
��3�������£�0.1mol?L-1NaHCO3��Һ��pH����8��c��OH-����c��H+����˵��HCO3-ˮ��̶ȴ��������̶ȣ���c��Na+����c��HCO3-����c��H2CO3����c��CO32-����ˮ��̶Ȳ�������c��HCO3-����c��H2CO3����c��HCO3-����c��OH-������Һ����������Դ��ˮ�ĵ�����HCO3-ˮ�⣬��c��OH-����c��H2CO3��������c��Na+����c��HCO3-����c��OH-����c��H2CO3����c��CO32-����
�ʴ�Ϊ��c��Na+����c��HCO3-����c��OH-����c��H2CO3����c��CO32-����
��1��pH=13��Ba��OH��2��Һ��c��OH-��=0.1mol/L�������������غ��֪����Һ��c[Ba��OH��2]=
c��OH-��=0.5��0.1mol/L=0.05mol/L���ʴ�Ϊ��0.05mol/L��
��2��pH=13��Ba��OH��2��Һ��������ʹ��ˮ��������ģ�ˮ���������c��H+��������Һ��ˮ�����c��OH -������ˮ�����c��OH -��=c��H+��=10-13mol/L��
�ʴ�Ϊ��10-13mol/L��
��3��pH=13��Ba��OH��2��Һ��c��OH-��=0.1mol/L����ijŨ��������Һ������ȣ�������֮�ȣ�1��9��Ϻٶ�����ֱ�Ϊ1L��9L��������ҺpH=11������Ϻ���Һ��c��OH -��=10-3mol/L���������Ũ��Ϊamol/L�����Ϻ���Һ��n��OH -��=1L��0.1mol/L-9L��amol/L=10-3mol/L����1L+9L�������a=0.01��
�ʴ�Ϊ��0.01mol/L��
����������ǿ����ʣ�������������ʣ���ͬpHֵ������ʹ�����Һ�ֱ��ˮϡ�ͣ�����ϡ�ʹٽ��������������������ӣ���ϡ����ͬ�ı����������PH����Ľ�С����ͼ��Ϊ����ϡ�͵�ͼ�ʴ�Ϊ����
��Һ̬SO2�ǹ��ۻ����ﲻ�ܵ��磬���ǵ���ʣ����ڷǵ���ʣ�
�۴���������ˮ�ܵ��磬�ǵ���ʣ�
��NaHCO3�����ӻ�����ǵ���ʣ�
��Ba��OH��2��Һ�ǻ��������ǵ����Ҳ���Ƿǵ���ʣ�
��ѡ���ڣ�
��1��NaHCO3�����Σ���ˮ��Һ����ȫ���룬����ǿ����ʣ��ʴ�Ϊ��ǿ��
��2��HCO3-ˮ������̼�������������ӣ�ˮ�����ӷ���ʽΪHCO3-+H2O

H2CO3+OH-���ʴ�Ϊ��HCO3-+H2O

H2CO3+OH-��
��3�������£�0.1mol?L-1NaHCO3��Һ��pH����8��c��OH-����c��H+����˵��HCO3-ˮ��̶ȴ��������̶ȣ���c��Na+����c��HCO3-����c��H2CO3����c��CO32-����ˮ��̶Ȳ�������c��HCO3-����c��H2CO3����c��HCO3-����c��OH-������Һ����������Դ��ˮ�ĵ�����HCO3-ˮ�⣬��c��OH-����c��H2CO3��������c��Na+����c��HCO3-����c��OH-����c��H2CO3����c��CO32-����
�ʴ�Ϊ��c��Na+����c��HCO3-����c��OH-����c��H2CO3����c��CO32-����
��1��pH=13��Ba��OH��2��Һ��c��OH-��=0.1mol/L�������������غ��֪����Һ��c[Ba��OH��2]=
| 1 |
| 2 |
��2��pH=13��Ba��OH��2��Һ��������ʹ��ˮ��������ģ�ˮ���������c��H+��������Һ��ˮ�����c��OH -������ˮ�����c��OH -��=c��H+��=10-13mol/L��
�ʴ�Ϊ��10-13mol/L��
��3��pH=13��Ba��OH��2��Һ��c��OH-��=0.1mol/L����ijŨ��������Һ������ȣ�������֮�ȣ�1��9��Ϻٶ�����ֱ�Ϊ1L��9L��������ҺpH=11������Ϻ���Һ��c��OH -��=10-3mol/L���������Ũ��Ϊamol/L�����Ϻ���Һ��n��OH -��=1L��0.1mol/L-9L��amol/L=10-3mol/L����1L+9L�������a=0.01��
�ʴ�Ϊ��0.01mol/L��
����������ǿ����ʣ�������������ʣ���ͬpHֵ������ʹ�����Һ�ֱ��ˮϡ�ͣ�����ϡ�ʹٽ��������������������ӣ���ϡ����ͬ�ı����������PH����Ľ�С����ͼ��Ϊ����ϡ�͵�ͼ�ʴ�Ϊ����

��ϰ��ϵ�д�
�����Ŀ
 �������� ��Cu ��Һ̬SO2 �۴����� ��NaHCO3 ��Ba��OH��2��Һ
�������� ��Cu ��Һ̬SO2 �۴����� ��NaHCO3 ��Ba��OH��2��Һ H2CO3+OH-
H2CO3+OH-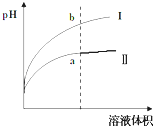 �������� ��Cu ��Һ̬SO2 �۴����� ��NaHCO3 ��Ba��OH��2��Һ
�������� ��Cu ��Һ̬SO2 �۴����� ��NaHCO3 ��Ba��OH��2��Һ HCO3-��H+��ƽ�ⳣ��K1= ������֪��10-5.60=2.5��10-6��
HCO3-��H+��ƽ�ⳣ��K1= ������֪��10-5.60=2.5��10-6��