��Ŀ����
ϴ����ȫ�������г���ȫ����Ҫ���ϡ�������������ײ��˲�䣬��ȫװ��ͨ����ʹ���еķ�ĩ�ֽ��ͷų������ĵ����γ����ң��Ӷ�����˾�����˿������˺���Ϊ�о���ȫ���ҹ����Ļ�ѧԭ����ȡ��ȫװ���еķ�ĩ����ʵ�顣����ɷ�����ȷ���÷�ĩ����Na��Fe��N��O����Ԫ�ء�ˮ������������������ĩ�����ܽ⡣����⣬������Ϊ������ף�������Ϊ����ɫ���壬���������ᡣ
ȡ13.0g������ף�����ʹ����ȫ�ֽ⣬���ɵ����͵����ң����ɵĵ����ۺϳɱ�״���µ����Ϊ6.72L���������ڸ��¸����������������벻�������ɫ��ĩ��Ӧ���ɻ������������һ�ֵ��ʡ�����������ڿ����п�ת��Ϊ�������κ�һ����̬���ʡ�
��ش��������⣺
��1���Ļ�ѧʽΪ �����ĵ���ʽΪ ��
��2�������ڿ�����ת��Ϊ̼�����Σ���Ӧ�Ļ�ѧ����ʽΪ ��
����̼����������ˮ�����Һ������Һ�и�����Ũ�ȴ�С��ϵΪ�� ��
��3�������������ɫ��ĩ������Ӧ�Ļ�ѧ����ʽΪ ����ȫ�����к���ɫ��ĩ�������� ��
��4�����������У��п�����Ϊ��ȫ�����к���ɫ��ĩ���Ʒ���� ��
ȡ13.0g������ף�����ʹ����ȫ�ֽ⣬���ɵ����͵����ң����ɵĵ����ۺϳɱ�״���µ����Ϊ6.72L���������ڸ��¸����������������벻�������ɫ��ĩ��Ӧ���ɻ������������һ�ֵ��ʡ�����������ڿ����п�ת��Ϊ�������κ�һ����̬���ʡ�
��ش��������⣺
��1���Ļ�ѧʽΪ �����ĵ���ʽΪ ��
��2�������ڿ�����ת��Ϊ̼�����Σ���Ӧ�Ļ�ѧ����ʽΪ ��
����̼����������ˮ�����Һ������Һ�и�����Ũ�ȴ�С��ϵΪ�� ��
��3�������������ɫ��ĩ������Ӧ�Ļ�ѧ����ʽΪ ����ȫ�����к���ɫ��ĩ�������� ��
��4�����������У��п�����Ϊ��ȫ�����к���ɫ��ĩ���Ʒ���� ��
| A��KCl | B��KNO3 | C��Na2S | D��CuO |
��1��NaN3��
(2)2N2O2+4CO2+2H2O=4NaHCO3+O2��c(Na+ )> c(HCO3�� ) > c(OH�� )> c(H+ )> c(CO32��)
(3)6Na+2Fe2O3=3Na2O2+4Fe , ��ɫ��ĩ�������dz䵱����������ȥNaN3�ֽ�����Ľ����ƣ�����������ˮ�����������Ⱥͼ����к����ʣ����ṩ��������������NaN3��Ѹ�ٷֽ⡣
��4��D.

(2)2N2O2+4CO2+2H2O=4NaHCO3+O2��c(Na+ )> c(HCO3�� ) > c(OH�� )> c(H+ )> c(CO32��)
(3)6Na+2Fe2O3=3Na2O2+4Fe , ��ɫ��ĩ�������dz䵱����������ȥNaN3�ֽ�����Ľ����ƣ�����������ˮ�����������Ⱥͼ����к����ʣ����ṩ��������������NaN3��Ѹ�ٷֽ⡣
��4��D.
��

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
 ���������⡣
���������⡣ �����ܽ���ˮ�Ƶã�����εĻ�ѧʽΪ ��
�����ܽ���ˮ�Ƶã�����εĻ�ѧʽΪ �� 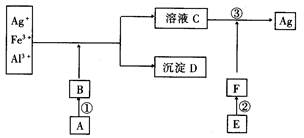
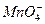
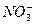 ��Cl-��
��Cl-��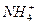
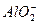 ��
��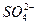
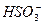
 �ŷŵĹ�
�ŷŵĹ� ҵ��ˮ�У�����K����Ag����Fe3����C1����OH����NO
ҵ��ˮ�У�����K����Ag����Fe3����C1����OH����NO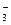 �������ӡ�
�������ӡ� ����д����Ԫ�ط��ţ���
����д����Ԫ�ط��ţ���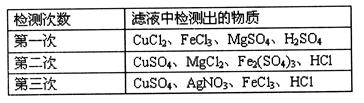
 ��
��