��Ŀ����
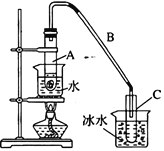
����Ŀ��ij��ѧС���������������������װ��������ͼ��ʾ�����û������Ʊ�����ϩ��
��֪��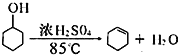
��Է������� | �ܶ�/g cm-3 | �۵�/�� | �е�/�� | �ܽ��� | |
������ | 100 | 0.96 | 25 | 161 | ������ˮ |
����ϩ | 82 | 0.81 | -103 | 83 | ������ˮ |
��1���Ʊ���Ʒ
��12.5 mL��������1mLŨ��������Թ�A�У�ҡ�Ⱥ�������Ƭ��������������Ӧ��ȫ�����Թ�C�ڵõ�����ϩ��Ʒ��
�����Թ��л�ϻ��Ѵ���Ũ�������ʱ������ҩƷ���Ⱥ�˳��Ϊ_________��
���������һ��ʱ��������Ǽ����Ƭ��Ӧ�ò�ȡ����ȷ������_________������ĸ����
A.�������� B.��ȴ�� C.���貹�� D.��������
�����Թ�C���ڱ�ˮ�е�Ŀ����_______________________________��
��2���Ʊ���Ʒ
������ϩ��Ʒ�к��л������������������ʵȡ����Ʒ�м��뱥��ʳ��ˮ�������á��ֲ㣬����ϩ��__________________��(��ϡ����¡�)����Һ����__________________������ĸ��ϴ����
a.����KMnO4��Һ b.ϡ���� c.Na2CO3��Һ
���ٽ��ᴿ��Ļ���ϩ����ͼ��ʾװ�ý�������ͼ������a ��������_______________��ʵ������ȴˮ��__________������ĸ���ڽ��롣����ʱҪ������ʯ�ң�Ŀ����__________________________��
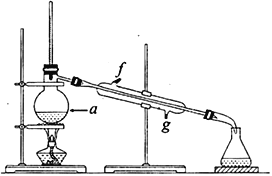
��3�������Ʊ���Ʒʱ���Ѵ����Ʒһ����������ʵ���ƵõĻ���ϩ��Ʒ����__________(�������������� ������)���۲�������ʵ�����õ��Ļ���ϩ����Ϊ6.25g���������___________________��
���𰸡� �Ƚ������������Թ�A�У��ٻ�������Ũ���� B ��ֹ����ϩ�Ļӷ�������������Ҳ������ �� c ������ƿ g ����ʣ���ˮ ���� 63.5%
��������(1)�ٸ����Ʊ����������IJ��������Թ��л�ϻ��Ѵ���Ũ�������ʱ��ҩƷ�ļ��뷽��Ϊ�Ƚ������������Թ�A�У��ٻ�������Ũ���ᣬ�ʴ�Ϊ���Ƚ������������Թ�A�У��ٻ�������Ũ���
���������һ��ʱ��������Ǽ����Ƭ����Ҫ��ȴ�ӣ���ѡB��
�ۻ���ϩ���۵�ܵͣ��е�Ҳ���ߣ����ӷ������Թ�C���ڱ�ˮ���Է�ֹ����ϩ�Ļӷ����ʴ�Ϊ����ֹ����ϩ�Ļӷ���
(2)�ٻ���ϩ������ˮ�����ܶȱ�ˮС�������á��ֲ��ϩ���ϲ㣻���ڷ�Һ��ϩ��Ʒ�л�������������ͻ������������Ʊ����������ᴿ����ʱ��c(Na2CO3��Һ)ϴ�ӿɳ�ȥ�ᣬ���������Ը�����أ��������������ϩ���ʴ�Ϊ���ϣ�c��
��ͼ������a ��������ƿ��Ϊ��ʹ������Ч�����ã���ȴˮ�������ܵ��¿ڼ�g�ڽ��룻��ʯ������ˮ��Ӧ�����������ƣ���ȥ�˲�����ˮ���õ������Ļ���ϩ���ʴ�Ϊ��������ƿ��g������ʣ���ˮ��
(3)�����Ʊ���Ʒʱ���Ѵ����Ʒһ�����������Ѵ����������½�����ʵ���ƵõĻ���ϩ��Ʒ�����������۲�����12.5 mL������������Ϊ12.5 mL��0.96 g cm-3=12g�����������ɻ���ϩ![]() ��82g/mol=9.84g������=
��82g/mol=9.84g������=![]() ��100%=63.5%���ʴ�Ϊ�����ڣ�63.5%��
��100%=63.5%���ʴ�Ϊ�����ڣ�63.5%��