��Ŀ����
���ͷ���һ�ֻ�ѧ���ɼ�������С�մ��ۣ�̼����泥�������[KAl(SO4)2?12H2O]�е�����������ɡ�ijС��Ϊ̽����ͬƷ�Ƶķ��ͷ۵Ļ�ѧ�ɷ֣���������ʵ�顣
��������衿
��1������1����С�մ�ͳ������
����2����С�մ���������
����3����__________________________���
�����������̡�
Ϊ̽����Ʒ�Ƶķ��ͷ۵ijɷ֣�ijͬѧ�������ʵ�飬�õ���������
��2����ϲ���١��۷���������AΪ________���÷��ͷ۵ijɷ�Ϊ__________________��
��3��������ٺ͢ڲ������䣨����Ҳ��ͬ�����������������ϡ�����Ϊ�����Ȼ�����Һ���۲쵽�а�ɫ�������ɣ��ܷ�ȷ�����ͷ۵ijɷֲ�˵�����ɣ�________________�� ____________________________________________________________________��
��4����Ʒ�Ƶķ��ͷ۵Ļ�ѧ��ɿ���Ϊ����2����������ʵ����֤��
ʵ����������Ʒ��ѡ����ѡ�Լ���ϡ���ᡢ0.1 mol/LNaOH��Һ��д��ʵ�鲽�衢Ԥ������ͽ��ۡ�
| ʵ�鲽�� | Ԥ������ͽ��� |
| ����1��ȡ������Ʒ��������������� ����Һ�ֳ����ݣ��ֱ�װ��A��B�Թ��С� | |
| ����2��_____________________________ __________________________________ | ________________________֤����Na+���� �ͷ�����NaHCO3�� |
| ����3��_____________________________ ___________________________________ | ___________________________________ _______����ϲ���2�еĽ��ۣ�����2������ |
��16�֣���1�����ۺ�������2�֣�
��2����������NH3����2�֣� NaHCO3��NH4HCO3��2�֣�д����Ҳ���֣�
��3�����ܣ�1�֣�����Ϊ����NaHCO3�ֽ��Na2CO3��Na2CO3������������BaC12��Һ��Ӧ���ɰ�ɫ��������2�֣� ע����˼�ӽ�Ҳ�÷�
��4����7�֣�����2���ýྻ�IJ�˿պȡA�е���Һ����
�ƾ������������գ��۲���ɫ��2�֣���ɫ�ʻ�ɫ��1�֣� ����3��������1����B�Թ�����εμ�
0.1 mol/L NaOH��Һ��2�֣������а�ɫ�������ɣ����Ȳ�����ɫ����������ܽ⣩��1�֣���֤�����ͷ�������������1�֣�
������2����պȡA�е���Һ����պȡB��Һ�����ھƾ������������գ�1�֣�������ɫ�ܲ����۲죨1�֣� ��ɫ����ɫ��1�֣���֤�����ͷ��к���������1�֣�
ע������2�У����ýྻ�IJ�˿պȡ����Һ����1�֣����ھƾ��ƻ��������ա���1�֡�
ֻд��պȡ����Һ���ø��ֵ㲻�÷֣����ھƾ��ơ��ƾ���ƻ��������ա������֡�
����3�У�������ɫ��Ӧ����������ϴ�Ӳ�˿������NaOH��Һ���飬��0.lmol/L NaOH
��Һ����1�֣�����εμӡ���1�֡�
���������������1�����������֪����ͬƷ�Ƶķ��ͷۿ���С�մ�ͳ�����ɣ�Ҳ������С�մ��������ɣ��������ɳ��ۺ�������ɣ�������֪�ļ���1������2�ƶϼ���3Ϊ���ۺ���������2������NaHCO3��NH4HCO3��Al2(SO4)2?12H2O����Ҫ���ʿ�֪��NH4HCO3����Σ�����ʯ�һ����ĥ���Էų���������������������ʯ����ĥ�����ܷų����壬��AΪNH3�����ͷۼ�һ�����г��ۣ�NH4HCO3�����ּ��Ⱥ���ȫ��Ϊ�����ݳ���NaHCO3���������ȱ�ΪNa2CO3�����CO2��H2O��Na2CO3������������Ӧ���ɶ�����̼���塢NaCl��H2O����Al2(SO4)2?12H2O����ˮ�����Al(OH)3�������϶�������������ɴ��ƶ�BΪCO2���÷��ͷ�һ������С�մ����Լ�Ʒ�Ƶķ��ͷ۵���Ҫ�ɷ���NaHCO3��NH4HCO3����3��Ba(OH)2�����������η�Ӧ��������ɫ�����ᱵ������Ҳ����̼���η�Ӧ�����ɰ�ɫ��̼�ᱵ��������˲���ȷ�����ͷ��к�����������С�մ�4������ʵ�鷽���в���2�Ľ������ƿ�֪������2���ýྻ�IJ�˿պȡA�е���Һ���پƾ������������գ��������ɫ�ʻ�ɫ��֤����Na+�����ͷ�����NaHCO3�����ڼ���2�Ƿ��ͷ���С�մ��������ɣ�����3�Ľ�����֤�����ͷ���Al2(SO4)2?12H2O�����������ʼ��ṩ�Լ��������ƶϣ�����3�����ʵ�鷽��֤�����ͷ��к���Al3+�����Ӧ��B�Թ�����εμ�0.1mol/LNaOH��Һ���۲쵽��ɫ�������Ȳ�����ɫ����������ܽ⣬֤����Al3+�����ͷ�����������
���㣺����̽��ʵ�鷽������Ƶ�֪ʶ��

 �ܿ�����ĩ��̾�ϵ�д�
�ܿ�����ĩ��̾�ϵ�д�ijУ��ѧС���ͬѧ������̼��Ϊ�缫����Ȼ�ͭ��Һʱ��������̼���ϳ����к�ɫ���������⣬����������ɫ����������Ϊ̽������̼���ϵIJ����������¹��̣�
�������
ͭ�Ļ�������ɫ�������£�
| ���� | ��ɫ������ | ���� | ��ɫ������ |
| ������ͭCu(OH)2 | ��ɫ���岻����ˮ | ����ͭ��CuSO4�� | ��Һ����ɫ |
| ������ͭ��CuO�� | ��ɫ���岻����ˮ | �Ȼ�ͭ��CuCl2�� | ��Һ����ɫ��ϡ��Һ����ɫ |
| �Ȼ���ͭ��CuCl�� | ��ɫ���岻����ˮ | ��ʽ�Ȼ�ͭ | ��ɫ���岻����ˮ |
��1��������裺�ٺ�ɫ����һ����ͭ����������Cu2O���ڰ�ɫ����Ϊͭ�Ļ�����仯ѧʽ����ΪCuCl
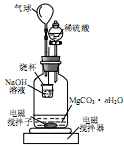
��2��ʵ����֤��ȡ���CuCl2��Һ�������̼����ϴ�ӡ������������װ�ý���ʵ�飬��֤��������

ʵ�������̼���ϵİ�ɫ���ʱ�Ϊ��ɫ����ˮ����ͭ����ɫ��
��̼���ϵĺ�ɫ�����Ƿ���Cu2O ����ǡ�����������
������cװ�ú�����ͨ�� �У������ְ�ɫ��������˵�����������еİ�ɫ����һ�����ڣ���д��װ��b�з�����Ӧ�Ļ�ѧ����ʽ��
��
��3���������ۣ�
�ٵ��CuCl2��Һ��������Ϸ����ķ�ӦΪ��Cu2++2e-��Cu�� ������ʯī�缫��ⱥ������ͭ��Һ���۲�����̼��������ֵ������ɫ���ʸ��ţ��ް�ɫ���ʣ����� �������⣬���ְ�ɫ���ʡ�
Ư����һ�ֳ��õ���������
��1����ҵ������Ư�۷�Ӧ�Ļ�ѧ����ʽΪ��________________ __��Ư�۵���Ч�ɷ�Ϊ ��
��2��ij̽��С����г��Ϲ�����һ����װ�����Ư�ۣ��Ը�Ư�۵ijɷֽ���̽�������������Լ������ʵ�鷽��������ʵ�顣���ڴ�������ʵ�鱨�档
��ѡ�Լ���2mol��L��1NaOH��Һ��2mol��L��1HCl��Һ��2mol��L��1HNO3��Һ��0.5mol��L��1BaCl2��Һ��0.01mol��L��1AgNO3��Һ������ʯ��ˮ��ʯ����Һ����̪��Һ������ˮ��
| ʵ�鲽�� | Ԥ����������� |
| ����1��ȡ����Ư��������������ˮ����ֽ��裬���ã����ˣ��ó�������Һ�� | |
| ����2���������������2mol��L��1HCl��Һ��������������ͨ�� | ���� ���ۣ� |
| ����3��ȡ��Һ��װA��B��֧�Թܡ���A�Թܣ� | ������Һ�ȱ��ɫ��Ȼ����ɫ�� ���ۣ� |
| ����4����B�Թܣ� | ��������ɫ������ ���ۣ� |
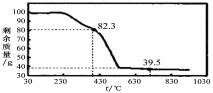
��3��̽��С��Ϊ�ⶨƯ����Ca(ClO)2�ĺ�������ȡƯ��bg��ˮ�ܽ�����Ƴ�100mL��Һ��ȷ��ȡ25.00mL����ƿ���������������KI��Һ����ַ�Ӧ����Һ�е��������0.1000mol/L��Na2S2O3��Һ�ζ����ζ�2�Σ�ƽ������Na2S2O3��Һ20.00mL�����Ư����Ca(ClO)2����������Ϊ_____________ _����ֻ����ʽ���������㣬��֪��Mr[Ca(ClO)2]="143" ��Ca(ClO)2+4HCl=2Cl2��+CaCl2+2H2O��2Na2S2O3+I2=Na2S4O6+2NaI��
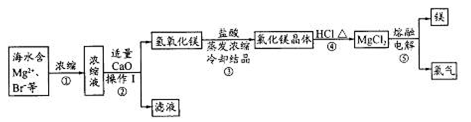
ijͭ��ʯ��ͭԪ�غ����ϵͣ��Һ�������þ���Ƶ��������ӡ�ijС����ʵ�������ý���-��ȡ���Ʊ�����ͭ��
��1������IΪ_______������II�õ��IJ����������ձ�_______
��2������II������III����ҪĿ����_______������ͭԪ�ء�
��3��С���Ա����CuSO4��Һ��Na2CO3��Һ��Ϸ�Ӧ���Ʊ�������ľ�ķ�����Cu2(OH)2CO3����Һ�����ʵ�鷢��������ɫ����Һ��ɫ���в��죬�������ϱ��������������������Ʋ�ͬʹ���л��н϶�Cu(OH)2��Cu4(OH)6SO4��
��֪Cu(OH)2��Cu2(OH)2CO3��Cu4((OH)6SO4��������ˮ����������ֽ��¶�����Ϊ 80�桢200�桢300�档
���ʵ���������Һ�ɷ֣���ɱ������ݡ�
��ѡ�Լ���2mol?L��1HCl��1 mol?L��1H2SO4��0.1 mol?L��1NaOH��0.1 mol?L��1 BaCl2������ˮ����������Ʒ��ѡ��
| ʵ�鲽�� | Ԥ������ͽ��� |
| ����1��ȡ��������Һ�����ˣ����ϴ�Ӻ�ȡ�������Թ��У�_______________________________________________________________ | ˵������Һ�л�__________________________________________,��Cu4( OH)6SO4�� |
| ����2����ȡ��������Һ���Թ��У�____________________________ | ______________�� ˵������Һ�л���Cu( OH) 2�� |
��4������ʵ����Ҫ100mL 0.5 mol?L��1��CuSO4��Һ������ʱ���ȡ_______gCuSO4?5H2O (��ѧʽ����250)��
ʵ�����Ʊ����ռ������SO2�������������¡�װ�ã�����SO2���������������Ӹ�������˳��Ϊ��ӣ� ���ӣ� ���ӣ� ���ӣ� ���ӣ棨��ӿ���ĸ�� �� ��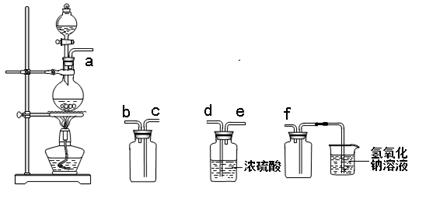
| A��b c d e | B��d e b c | C��d e c b | D��e d b c |
������Ũ���������ü�ʯ�Ҹ�����ǣ� ��
| A��Cl2 | B��CO | C��SO2 | D��NH3 |