��Ŀ����
����Ŀ����������ֲ�������������ҪԪ�ء��ش��������⣺
��1������Nԭ�ӵ����Ų�ͼ��ʾ��״̬�У������ɵ͵��ߵ�˳����____�����ţ���
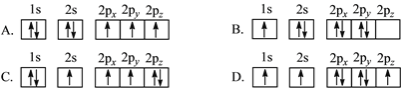
��2���ǰ���NH2OH���ɿ����ǰ������ڵ�һ����ԭ�ӱ��ǻ�ȡ���������������N���ӻ�������___���ǰ�������ˮ������Ҫԭ����____��
��3����һ������I1(N)_____I1(P)�����������������������ԭ����_____��
��4��ֱ�������������������������������������������ͨ�����ö�����ԭ�����������ģ���ͼ��ʾ������n�������������γɵ�������������ӵ�ͨʽΪ_____��
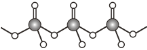
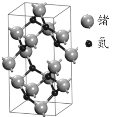
��5�������������ʴ��Ӳ�ȸߵ��ŵ㣬��������ԭ���뵪ԭ��֮��������Ե�s-p�ӻ��������ྦྷ������______���塣һ�ֵ����ྦྷ�������ģ����ͼ���仯ѧʽΪ_____��������������ĸ�Ϊbpm�������ӵ�����ֵΪNA��������ܶ�Ϊ�� g/cm3�������������εı߳�Ϊ_______ pm���г�����ʽ����
���𰸡�A��C��B��D sp3�ӻ� �ǰ�������ˮ���Ӿ�Ϊ���Է��ӣ����Ӽ䶼���γ���� �� N��ԭ�Ӱ뾶��PС���˶��������ӵ��������ϴ� [PnO3n+1](n+2)- ԭ�� Ge3N4 ![]() ��1015
��1015
��������
(1)ԭ�Ӻ�������Ų��У����������ռ�Ĺ���ܼ�Խ�ߣ���ԭ������Խ�ߣ��ݴ��жϣ�
(2)������Nԭ����3��ԭ������������1���µ��Ӷԣ��ǰ�������ˮ���Ӿ�Ϊ���Է��ӣ����Ӽ䶼���γ�������ݴ˽��
(3)����ԭ�Ӱ뾶�Ĵ�С����Ϻ˶��������ӵ������������жϣ�
(4)����n����ԭ�ӵĶ����������ӣ��൱����n�������������ȥ����(n-1)��ԭ�ӣ����Եó�P��Oԭ�ӹ�ϵ�����ϼ۴����͵�������������ɣ�
(5)���ݵ����������ʴ��Ӳ�ȸߵ��ŵ��жϾ������ͣ����ݷ�̯�����㾧���к��е���ԭ�Ӻ͵�ԭ����Ŀ���ݴ���д������Ļ�ѧʽ������ܶȵļ��㹫ʽ�������
(1)ԭ�Ӻ�������Ų��У����������ռ�Ĺ���ܼ�Խ�ߣ���ԭ������Խ�ߣ����������1s��2s��2p������ռ��2p������ӵ���Ŀ��֪��A������ͣ�D������ߣ�����ռ��2s������ӵ���Ŀ��֪������B��C����������ɵ͵��ߵ�˳����A��C��B��D���ʴ�Ϊ��A��C��B��D��
(2)�ǰ�(NH2OH)�ɿ����ǰ������ڵ�һ����ԭ�ӱ��ǻ�ȡ���������������Nԭ����3��ԭ������������1���µ��Ӷԣ�����sp3�ӻ����ǰ�������ˮ������Ϊ�ǰ�������ˮ���Ӿ�Ϊ���Է��ӣ����Ӽ䶼���γ�������ʴ�Ϊ��sp3�ӻ����ǰ�������ˮ���Ӿ�Ϊ���Է��ӣ����Ӽ䶼���γ������
(3)N��ԭ�Ӱ뾶��PС���˶��������ӵ��������ϴ���˵�һ������I1(N)��I1(P) ���ʴ�Ϊ������N��ԭ�Ӱ뾶��PС���˶��������ӵ��������ϴ�
(4)����n��Pԭ�ӵĶ����������ӣ��൱����n�������������ȥ����(n-1)��ԭ�ӣ�Oԭ����Ŀ=3n-(n-1)=3n+1���������Ϊ(-2)��(3n+1)+5n=-(n+2)������n�������������γɵ�������������ӵ�ͨʽΪ��[PnO3n+1](2+n)-���ʴ�Ϊ��[PnO3n+1](2+n)-��
(5)�����������ʴ��Ӳ�ȸߵ��ŵ㣬����ԭ�Ӿ������������˵���������ԭ�Ӿ��壻�����к��е���ԭ����Ŀ=10��![]() +4��
+4��![]() =6����ԭ����Ŀ=8����˵�����Ļ�ѧʽΪGe3N4��
=6����ԭ����Ŀ=8����˵�����Ļ�ѧʽΪGe3N4��
�辧�����������εı߳�Ϊx pm����Ϊ����������=![]() g�����������=x2b pm3������ g/cm3=
g�����������=x2b pm3������ g/cm3=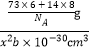 �����x2=
�����x2=![]() ��1030�����x=
��1030�����x=![]() ��1015���ʴ�Ϊ��
��1015���ʴ�Ϊ��![]() ��1015��
��1015��

����Ŀ��һ���¶�ʱ�����ݻ�Ϊ 2 L ���ܱ������г���һ������ SO2(g)�� O2(g)��������Ӧ��2SO2(g)+ O2(g)2SO3(g) ��H = - 196 kJ/mol��һ��ʱ���Ӧ�ﵽƽ��״̬����Ӧ�����вⶨ�IJ������������ʾ��
��Ӧʱ��/min | n(SO2)/mol | n(O2)/mol |
0 | 2 | 1 |
5 | 1.2 | |
10 | 0.4 | |
15 | 0.8 |
����˵������ȷ����
A. ǰ 5 min ��ƽ����Ӧ����Ϊ��(SO2) = 0.08 mol/(L��min)
B. �����¶Ȳ��䣬��ƽ�����������ٳ��� 0.2 mol SO2(g)�� 0.2 mol SO3(g)ʱ������ > ����
C. ����������������������ʼʱ�������г��� 2 mol SO3(g)���ﵽƽ��״̬ʱ���� 78.4 kJ������
D. ��ͬ�¶�������ʼʱ�������г��� 1.5 mol SO3(g)���ﵽƽ��״̬ʱ SO3 ��ת����Ϊ 40%