��Ŀ����
����Ŀ��ʵ������Ҫ0.1 mol��L��1 NaOH��Һ450 mL��0.5 mol��L��1������Һ500 mL��������������Һ����������ش��������⣺
��1������ͼ��ʾ��������������Һ�϶�����Ҫ����____________________(����ĸ)��
������������Һ�����õ��IJ���������__________________(����������)��
�������й�����ƿʹ��˵������ȷ������ ��
a������ƿʹ��ǰһ��Ҫ����Ƿ�©Һ�������Ϊװˮ����ƿ���������۲��Ƿ�©ҹ����������������ת180����������۲��Ƿ�©ҹ��
b��������Ϊ��Ӧ��������������Һ�����������ǿ������������������Һ��
c�����ܽ������Ũ��Һֱ��������ƿ���ܽ��ϡ�ͣ����ܼ���������ȵ�Һ�� ��
d��������ƿ������Һ������ʱ���ӿ̶��ߣ�������ҺŨ��ƫС��
(2)������NaOH��Һʱ��
�ٸ��ݼ�����������ƽ��ȡNaOH������Ϊ________g��
����NaOH��Һ��ת��������ƿʱ����������������������ҺŨ��___________(�>������<������)0.1 mol��L��1��
����NaOH�����ܽ��������������ƿ��ϴ���ձ�����������ϴ��Һ��������ƿ�����ݣ���������ҺŨ��___________(�>������<������) 0.1 mol��L��1��
(3)������������Һʱ��
��������������Ϊ98%���ܶ�Ϊ1.84 g��cm��3��Ũ��������Ϊ__________(����������һλС��)mL��
�����ʵ������15 mL��50 mL��100 mL��Ͳ��Ӧѡ��___________mL��Ͳ��ã�
�����ƹ������������ձ��н�Ũ�������ϡ�ͣ�ϡ��ʱ����������__________________________��
���𰸡�(1)AC �ձ��������� bd
(2)��2.0��2�֣� ��<��2�֣� ��>��2�֣�
(3)��13.6��2�֣� ��15��2�֣� �۽�Ũ���������ڻ�������ˮ�У����ò��������Ͻ�����2�֣�
��������
�����������1����������Һ��Ҫ���ֲ�����������Ͳ���ձ�������������ͷ�ιܡ�����ƿ������Ҫ������ƿ�ͷ�Һ©����
���������Ϸ�����֪����Ҫ���������ձ��Ͳ�������
��a������ƿʹ��ǰһ��Ҫ����Ƿ�©Һ�������Ϊװˮ����ƿ���������۲��Ƿ�©ҹ����������������ת180����������۲��Ƿ�©ҹ��a��ȷ��b��������Ϊ��Ӧ��������������Һ��������ֻ������һ���������Һ��b����c�����ܽ������Ũ��Һֱ��������ƿ���ܽ��ϡ�ͣ����ܼ���������ȵ�Һ����c��ȷ��d��������ƿ������Һ������ʱ���ӿ̶��ߣ�������ҺŨ��ƫ��d����ѡbd��
��2��������450 mL������ƿ������NaOH��ҺҪ��500 mL������ƿ��m��NaOH��=cVM=0.1 molL-1��0.5 L��40 gmol-1=2.0 g��
����NaOH��Һ��ת��������ƿʱ����������������������ҺŨ��ƫС����С��0.1mol/L��
��NaOH����ˮ�ų������ȣ�Ӧ������ȴ�����º�����������ƿ�У������ݺ���Һ��ȴ�����º������С��Ũ��ƫ�ߣ�
��3����c��Ũ��V��Ũ��=c��ϡ��V��ϡ������1000��1.84��98%/98��V��Ũ��=0.5��0.5������V��Ũ����0.013 6 L=13.6 mL����ѡ��15 mL��Ͳ��ã����С����Ũ��������ˮ���ȣ����ܶȴ���ˮ������ϡ��Ũ����IJ���Ϊ����Ũ���������ڻ�������ˮ�У����ò��������Ͻ�����

����Ŀ��A��B��C��D����������ˮ�Ļ�����ֻ�ɱ��еİ���������ɣ������ֻ����������������Ӹ�����ͬ��
������ | Cl����SiO32-��OH����NO3- |
������ | H����Ag����Na����K�� |
��֪��A��Һ��ǿ���ԣ���A��Һ��B��C��Һ��Ͼ�������ɫ������B����ɫ��Ӧ�ʻ�ɫ��
�ش��������⣺
��1��д��A��B��C��D�Ļ�ѧʽ��A______��B_____��C____��D____��
��2��д��A��Һ��B��Һ��Ӧ�����ӷ���ʽ�� __________��
��3����ѡ���ʺϴ��A��C��D��Һ���Լ�ƿ����ţ�
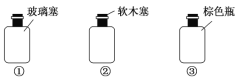
��Һ | A | C | D |
�Լ�ƿ��� | ___ | ___ | ___ |
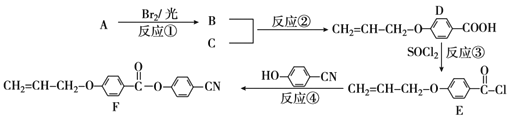
����Ŀ����ͼ��ʾװ���й۲쵽������ָ��ƫת��M����֡�N����ϸ���Դ��ж��±�������M��N��P���ʣ����п��Գ�������
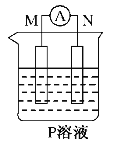
ѡ�� | M | N | P |
A | п | ͭ | ϡ������Һ |
B | ͭ | �� | ϡ������Һ |
C | �� | п | ��������Һ |
D | п | �� | ��������Һ |
A.AB.BC.CD.D