��Ŀ����
20��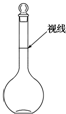 ʵ��������500mL 0.2mol•L-1��Na2SO4��Һ��ʵ����������У�
ʵ��������500mL 0.2mol•L-1��Na2SO4��Һ��ʵ����������У�A������ƽ�ϳƳ�14.2g�����ƹ��壬���������ձ��У�������������ˮʹ����ȫ�ܽⲢ��ȴ�����£�
B�����Ƶõ���ҺС�ĵ�ת�Ƶ�����ƿ�У�
C������������ƿ�м�����ˮ��Һ���̶���1��2cm�������ý�ͷ�ι�С�ĵμ�����ˮ����Һ��Һ����ʹ���̶������У�
D������������ˮϴ���ձ��Ͳ�����2��3�Σ�ÿ��ϴ�ӵ�Һ�嶼С��ע������ƿ����������
E��������ƿƿ���������������µߵ���ҡ�ȣ�
����д���пհף�
��1�������������ȷ˳��Ϊ������ţ�ABDCE��
��2����ʵ���õ��Ļ������������ձ�����ƽ�������롢���ӣ�������������ȱ�ٵ���������Ͳ��500ml����ƿ����ͷ�ιܣ�
��3�����������ʹ������ҺŨ��ƫ�ߵ��ǣ�����ţ�ad��
a��ijͬѧ�۲�Һ��������ͼ��ʾ
b��û���������IJ�������D
c��������ˮʱ�����������˿̶���
d��������մ������
e������ƿʹ��ǰ�ڱ�մ��ˮ�飮
���� ��1������������Һ��ʵ��������̽��в������������
��2������������Һ��ʵ���������ѡ������������
��3�������������������ʵ����ʵ�������Һ�������Ӱ�죬����C=$\frac{n}{V}$������������
��� �⣺��1������500ml0.2mol/L��Na2SO4��Һ�����������м��㡢�������ܽ⡢��Һ��ϴ����Һ�����ݡ�ҡ�ȵȲ�������������ƽ��������ҩ��ȡҩƷ�����ձ����ܽ⣨������Ͳ��ˮ�����ò��������裬�����ܽ⣬�ָ������º�ת�Ƶ�500mL����ƿ�У����ò�����������ϴ��2-3�Σ���ϴ��Һת�Ƶ�����ƿ�У���ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ�����ݵߵ�ҡ�ȣ���˳��ΪABDCE��
�ʴ�Ϊ��ABDCE��
��2������500ml0.2mol/L��Na2SO4��Һ�����������м��㡢�������ܽ⡢��Һ��ϴ����Һ�����ݡ�ҡ�ȵȲ������õ��������У�������ƽ���ձ�������������Ͳ��500mL����ƿ����ͷ�ιܣ�
��ȱ�ٵ������У���Ͳ��500ml����ƿ����ͷ�ιܣ�
�ʴ�Ϊ����Ͳ��500ml����ƿ����ͷ�ιܣ�
��3��a��ijͬѧ�۲�Һ��������ͼ��ʾ��������Һ�棬����Һ���ƫС��Ũ��ƫ�ߣ���a��ȷ��
b��û���������IJ�������D���������ʵ���ʧ����Ũ��ƫ�ͣ���b����
c��������ˮʱ�����������˿̶��ߣ�����Һ���ƫ��Ũ��ƫС����c����
d��������մ�����ʣ������������ƫ��������ҩƷ������ƫ����Ũ��ƫ�ߣ���d��ȷ��
e������ƿʹ��ǰ�ڱ�մ��ˮ�飬��Ũ����Ӱ�죬��e����
��ѡad��
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƣ���Ϥ���ƹ����ǽ���ؼ���ע����������������Ŀ�ѶȲ���

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | X����ijЩ�ǽ���Ԫ�ػ��� | |
| B�� | X����Ϊ����Ԫ�� | |
| C�� | Xԭ�ӵ������������ͺ˵�����϶�Ϊ���� | |
| D�� | X�����γɻ�ѧʽΪKXO3���� |
| A�� | ����ȼ��þ���������CO2����ļ���ƿ�У�2Mg+CO2�T��ȼ 2MgO+C | |
| B�� | ����ʯ���ڴ��CO32-+2CH3COOH�T2CH3COO-+CO2��+H2O | |
| C�� | ��NH4HSO4ϡ��Һ����μ���Ba��OH��2ϡ��Һ��SO42-�պó�����ȫ��Ba2++2OH-+NH4++H++SO42-�TBaSO4��+NH3•H2O+H2O | |
| D�� | �����KI��Һ�еμ�ϡ���ᣬ�ڿ����з���һ��ʱ�����Һ������4H++4I-+O2�T2I2+2H2O |
| A�� | Zn��s��+H2SO4��aq���TZnSO4��aq��+H2����g����H��0 | B�� | 2CO��g��+O2��g���T2CO2��g����H��0 | ||
| C�� | C��s��+CO2��g���T2 CO��g����H��0 | D�� | H+��aq��+OH-��aq���TH2O ��l����H��0 |
| A�� | ��С�������ݻ� | B�� | ʹ�ô��� | C�� | ��ѹ�³���He | D�� | �����³���Cl2 |
| A�� | ��Na2CO3+SiO2$\frac{\underline{\;����\;}}{\;}$Na2SiO3+CO2����֪������H2SiO3��H2CO3 | |
| B�� | �������Ҫ�ܷ����������IJ����Լ�ƿ�� | |
| C�� | ���������Һ�м������������ɫ����������ʱ�����ܽ� | |
| D�� | ��������������������������Ϊ�ڻ�NaOH�����װ�� |
| A�� | C2H6 | B�� | C4H10 | C�� | C5H12 | D�� | C7H16 |
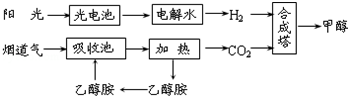
��1���úϳ�·�߶��ڻ��������ļ�ֵ���ڼ����̵����ж�����̼�ŷŵ��������������ЧӦ��
��2��15��20%���Ҵ�����HOCH2CH2NH2��ˮ��Һ���������ԣ������ϳ���·������CO2���ռ��������ӷ���ʽ��ʾ�Ҵ���ˮ��Һ�������Ե�ԭ��HOCH2CH2NH2+H2O?HOCH2CH2NH3++OH-����
��3��CH3OH��l����H2��ȼ���ȷֱ�Ϊ����H=-725.5kJ/mol����H=-285.8kJ/mol��д����ҵ����CO2��H2�ϳ�CH3OH���Ȼ�ѧ����ʽ��CO2��g��+3H2��g���TCH3OH��l��+H2O��l����H=-131.9KJ/mol��
��4���ݱ�������300�桢70MPa���ɶ�����̼�������ϳ��Ҵ�Ҳ�ѳ�Ϊ��ʵ��
2CO2��g��+6H2��g����CH3CH2OH��g��+3H2O��g��������������ȷ����acd��
a��ʹ�ô������ܴ���������Ч��
b����Ӧ����300����п��Ʋ�÷�Ӧ�����ȷ�Ӧ
c���������CO2��������H2��ת����
d����ƽ���������з����CH3CH2OH��H2O�����CO2��H2��������
��5����һ��������ܱ������У�CO2��H2Ҳ���Խ������»�ѧ��Ӧ��
CO2��g��+H2��g�� CO��g��+H2O��g����
�仯ѧƽ�ⳣ��K���¶�T�Ĺ�ϵ�����ʾ��
| T/�� | 700 | 800 | 830 | 1000 | 1200 |
| K | 0.6 | 0.9 | 1.0 | 1.7 | 2.6 |
�ٸ÷�ӦΪ���ȷ�Ӧ������ȡ����ȡ�����
�����жϸ÷�Ӧ�ﵽ��ѧƽ��״̬��������BC��
A��������ѹǿ���� B�����������c��CO������ C��v��H2����=v��H2O����
D��c��CO��=c��H2�� E�����������ܶȱ��ֲ��� F����������ƽ����Է����������ֲ���
����800��ʱ������������Ӧ��ijһʱ�̲�������ڸ����ʵ�Ũ�ȷֱ�Ϊc��CO2��Ϊ2mol/L��c��H2��Ϊ 1.5mol/L��c��CO�� Ϊ 1mol/L��c��H2O�� Ϊ 3mol/L������һʱ�̣���Ӧ�������ƶ������������������
