��Ŀ����
����Ŀ��ijϡ�����ϡ����Ļ����Һ200 mL��ƽ���ֳ�����.������һ��������ͭ�ۣ��������19.2 g(��֪����ֻ����ԭΪNO����)������һ�����������ۣ�������������������������ӵı仯��ͼ��ʾ�����з�����������ȷ����(����)
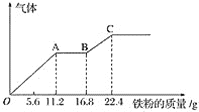
A.ͼ�У�AB�εķ�ӦΪFe��2Fe3��=3Fe2������Һ����������Ϊ��������
B.ԭ��Һ������Ũ��Ϊ2.5 mol��L��1
C.ԭ�����Һ����������ӵ����ʵ���Ϊ0.4 mol
D.ͼ�У�OA�β�����������һ��������BC�β���������������
���𰸡�B
��������
A. OA�κ�BC�ζ����������ɣ���AB��û���������ɣ��������̷����ķ�Ӧ��Fe��4H����NO3-=Fe3+��NO����2H2O��2Fe3����Fe=3Fe2����Fe��2H��=Fe2����H2��������AB�η�����Ӧ2Fe3����Fe=3Fe2������A��ȷ��
B. ��ͼ����Կ�������ȫ��Ӧʱ����Ӧ����Fe��������22.4 g(0.4 mol)������������ʵ���Ҳ��0.4 mol(��Ϊ������Һ��ֻ��FeSO4)����Һ�����100 mL����Ũ����4 mol��L-1����B����
C. ��һ�ݼ���ͭ�ۣ�������Ӧ��3Cu��8H����2NO3-=3Cu2����2NO����4H2O��Cu�����ʵ�����0.3 mol����һ����Һ���������0.2 mol�������ܵ��������0.4 mol����C��ȷ��
D. OA�κ�BC�ζ����������ɣ���AB��û���������ɣ��������̷����ķ�Ӧ��Fe��4H����NO3-=Fe3+��NO����2H2O��2Fe3����Fe=3Fe2����Fe��2H��=Fe2����H2��������AB�η�����Ӧ2Fe3����Fe=3Fe2����OA�η�����ӦFe��4H����NO3-=Fe3+��NO����2H2O����BC�η�����ӦFe��2H��=Fe2����H2������D��ȷ��
��ĿҪ��ѡ����ȷ�ģ���ѡB��

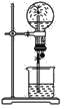
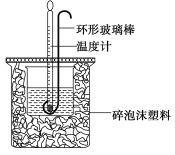
����Ŀ��ʵ������50 mL 0.50 mol��L��1���ᡢ50 mL 0.55 mol��L��1 NaOH��Һ����ͼ��ʾװ�ý��вⶨ�к��ȵ�ʵ�飬�õ����е����ݣ�
ʵ����� | ��ʼ�¶�t1/�� | ��ֹ�¶�t2/�� | |
���� | NaOH��Һ | ||
1 | 20.2 | 20.3 | 23.7 |
2 | 20.3 | 20.5 | 23.8 |
3 | 21.5 | 21.6 | 24.9 |
������������⣺
��1��ʵ��ʱ�û��β�����������Һ�ķ�����____________________��
������ͭ˿��������滷�β�������������_____________________��
��2�������ݴ�����t2��t1��3.4 �档���ʵ���õ��к��Ȧ�H��________[�����NaOH��Һ���ܶȰ�1 g��cm��3���㣬��Ӧ������Һ�ı�����(c)��4.18 J��(g����)��1����]��
��3������NaOH��Һ��Ϊ��ͬ�������ͬŨ�ȵİ�ˮ������к���Ϊ��H1����H1�릤H�Ĺ�ϵΪ����H1__________��H(�����������������)��������_______________��
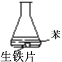
����Ŀ�����ж�����Ƭ��ʴ�Ա�ʵ��ķ�����ȷ���ǣ� ��
��� | �� | �� | �� |
ʵ�� |
|
|
|
���� | 8Сʱδ�۲� ��������ʴ | 8Сʱδ�۲� ��������ʴ | 1Сʱ�۲� ��������ʴ |
A.�Ա�ʵ��٢ڢۣ�˵�����ܸ���O2
B.ʵ����У�����Ƭδ��������ʴ����Ҫԭ����ȱ��O2
C.ʵ����У�NaCl��Һ���ܽ��O2������ʹ����Ƭ������ʴ
D.ʵ����У���������������ʴ��������Ӧ��O2+4e-+2H2O=4OH-