��Ŀ����
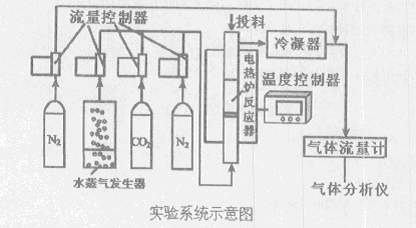
ú��ֱ��ȼ�����������ص���̬�ͻ�����Ⱦ��������ֱ��ȼ�պ��ѽ����������������⡣���ú̿������Ч�����������й���չ��̼���õĹؼ�����ͼ��ú�����ͳ����ʵ��ϵͳʾ��ͼ��
�����������
I��������ʵ���¶ȣ�ͨ�˸ߴ�N2����Ͷ�Ͽڼ���ʯӢɰ���������壩
��ֹͣͨ��N2���ӷ�Ӧ���·�������ͨ��ˮ������CO2����Ͷ�Ͽڼ���CaO
��ֹͣͨ��ˮ������CO2����Ͷ�Ͽڼ���ú
��������ͨ��ˮ������CO2����ʼ��Ӧ����������ڹ���ͨ�˸ߴ�N2
��1���������������� ��
��2����ʹ��CaCO3����CaO����CaCO3����Ļ�ѧ����ʽΪ ��
��3��ͨ��N2��Ŀ�ģ� ��
��4����Ӧ���з������¼��ַ�Ӧ��
C��s��ʮH2O��g��=CO��g��+H2��g�� ��H=+131kJ/mol
C��s��+O2��g��=CO2��g�� ��H=-394kJ/mol
CO��g��+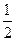 O2��g��=CO2��g�� ��H=-283kJ/mol
O2��g��=CO2��g�� ��H=-283kJ/mol
CO��g��+H2O��g��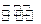 H2��g��+CO2��g��
H2��g��+CO2��g��
��ͼ�ܱ�ʾCO��H2O��g������CO2��H2�ķ�Ӧ���̺�������ϵ��ͼ�� ������ţ���
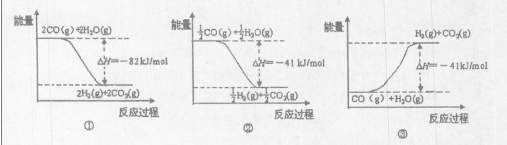
��5����֪830��ʱ��CO��g��+H2O��g��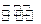 H2��g��+CO2��g����ƽ�ⳣ��K=1�������ʵ�����CO��H2O��Ӧ�ﵽƽ��ʱ��CO��ת����Ϊ .
H2��g��+CO2��g����ƽ�ⳣ��K=1�������ʵ�����CO��H2O��Ӧ�ﵽƽ��ʱ��CO��ת����Ϊ .
��6��ú���������л���������CH4����֪CH4��CaSO4��Ӧ����CaS��CO2��H2O���÷�Ӧ���������뻹ԭ�������ʵ���֮��Ϊ ��
��7��ú�������������CO��H2��CH4����ɣ���������ȼ�ϵ�ء����м���������CH4���뷴Ӧ�ĵ缫��ӦʽΪ ��

�����������
I��������ʵ���¶ȣ�ͨ�˸ߴ�N2����Ͷ�Ͽڼ���ʯӢɰ���������壩
��ֹͣͨ��N2���ӷ�Ӧ���·�������ͨ��ˮ������CO2����Ͷ�Ͽڼ���CaO
��ֹͣͨ��ˮ������CO2����Ͷ�Ͽڼ���ú
��������ͨ��ˮ������CO2����ʼ��Ӧ����������ڹ���ͨ�˸ߴ�N2
��1���������������� ��
��2����ʹ��CaCO3����CaO����CaCO3����Ļ�ѧ����ʽΪ ��
��3��ͨ��N2��Ŀ�ģ� ��
��4����Ӧ���з������¼��ַ�Ӧ��
C��s��ʮH2O��g��=CO��g��+H2��g�� ��H=+131kJ/mol
C��s��+O2��g��=CO2��g�� ��H=-394kJ/mol
CO��g��+
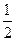 O2��g��=CO2��g�� ��H=-283kJ/mol
O2��g��=CO2��g�� ��H=-283kJ/molCO��g��+H2O��g��
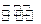 H2��g��+CO2��g��
H2��g��+CO2��g����ͼ�ܱ�ʾCO��H2O��g������CO2��H2�ķ�Ӧ���̺�������ϵ��ͼ�� ������ţ���

��5����֪830��ʱ��CO��g��+H2O��g��
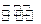 H2��g��+CO2��g����ƽ�ⳣ��K=1�������ʵ�����CO��H2O��Ӧ�ﵽƽ��ʱ��CO��ת����Ϊ .
H2��g��+CO2��g����ƽ�ⳣ��K=1�������ʵ�����CO��H2O��Ӧ�ﵽƽ��ʱ��CO��ת����Ϊ .��6��ú���������л���������CH4����֪CH4��CaSO4��Ӧ����CaS��CO2��H2O���÷�Ӧ���������뻹ԭ�������ʵ���֮��Ϊ ��
��7��ú�������������CO��H2��CH4����ɣ���������ȼ�ϵ�ء����м���������CH4���뷴Ӧ�ĵ缫��ӦʽΪ ��

��

��ϰ��ϵ�д�
�����Ŀ
 ��
��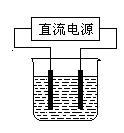
 2H2����O2�����������Ǹ�ԭ�������������Һ��С�������� g��
2H2����O2�����������Ǹ�ԭ�������������Һ��С�������� g��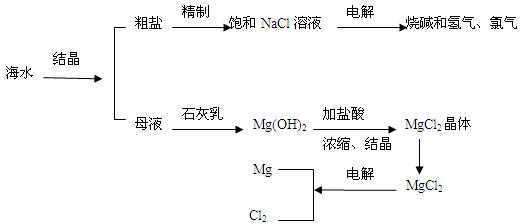
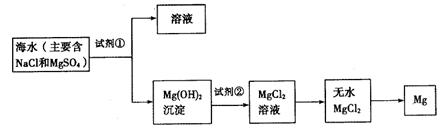
 ��һ���ں�ˮ��þ�Ĺ��������ʵ�ֶ�þ���ӵĸ���
��һ���ں�ˮ��þ�Ĺ��������ʵ�ֶ�þ���ӵĸ���