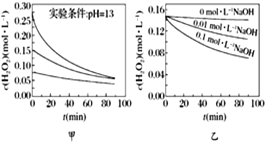
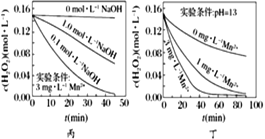
��Ŀ����
����Ŀ�����������£�����������5L�ܱ������У�����2molCO��3molH2������Ӧ��CO(g)+2H2(g)![]() CH3OH(g)��ƽ���CH3OH�����ʵ���Ϊ1mol������������ȷ����
CH3OH(g)��ƽ���CH3OH�����ʵ���Ϊ1mol������������ȷ����
A. �淴Ӧ�Ľ��У������ƽ����Է���������С
B. H2��ƽ��ת����Ϊ50%
C. ���¶ȷ�Ӧ��ƽ�ⳣ��K��25L2��mol��2
D. ƽ�������������ͨ��3mol CO��ƽ�ⲻ�ƶ�
���𰸡�C
��������
CO(g)+2H2(g)![]() CH3OH(g)
CH3OH(g)
��ʼ��(mol) 2 3 0
ת����(mol) 1 2 1
ƽ����(mol) 1 1 1
A����ΪM=m/n���淴Ӧ�Ľ��У������������������䣬�����ܵ����ʵ�����С���ʻ�������ƽ����Է�������������A����
B��������ƽ��ת����Ϊ2/3��100%��66.7%����B����
C�����¶ȷ�Ӧ��ƽ�ⳣ��K=![]() ��25����C��ȷ��
��25����C��ȷ��
D��ƽ�������������ͨ��3molCO����Ӧ��Ũ������ƽ�������ƶ�����D����
��ѡC��

����Ŀ��������Դ�����þ��й���ǰ����
��1����ͼ�ǴӺ�ˮ����ȡþ�ļ����̡�
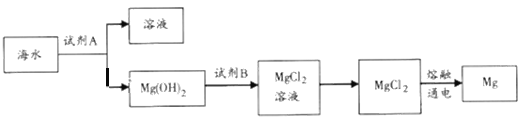
�ٹ�ҵ�ϳ����ڳ���Mg2+���Լ�A��__________���ѧʽ����Mg(OH)2ת��ΪMgCl2�����ӷ���ʽΪ________________________��
������ˮMgCl2��ȡMg�Ļ�ѧ����ʽ��______________________��
��2���������и�����I����ʽ���ڵĵ�Ԫ�أ�ʵ������ȡI2��;��������ʾ��
![]()
�����պ������ҽ�ʱ���õ���Ҫ������__________�������ƣ���
�����ữ����Һ�мӹ���������Һ���÷�Ӧ�����ӷ���ʽΪ__________��
�۷�Ӧ��������CCl4����ȡ��,������ȡ����Һ�ķ����ӵ�ˮ����ȡ�⣬��Ҫ������������ͼ��
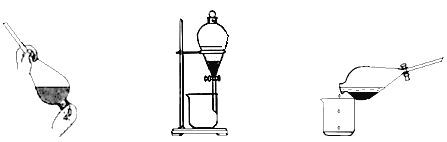
��.������ȡ������ ��.���÷ֲ� ��.����
�ס��ҡ���3��ʵ������У��������__________������������������������������
��3����ˮ�в������ӵĺ�������:
�ɷ� | ������mg/L�� | �ɷ� | ������mg/L�� |
Na+ | 10560 | Cl�� | 18980 |
Mg2+ | 1272 | Br�� | 64 |
Ca2+ | 400 | SO42�� | 2560 |
����100L�ú�ˮ����ȡþ��������������Լ�A__________g��
����Ŀ��ToC�£������Ϊ2 L�ĺ����ܱ�������ͨ��NO2��O2��������Ӧ��4NO2(g)+O2(g)![]() 2N2O5(g)��H��0������ʵ���������±�������˵������ȷ����
2N2O5(g)��H��0������ʵ���������±�������˵������ȷ����
ʱ��/s | 0 | 5 | 10 | 15 |
n(NO2)/mol | 8.00 | n1 | n2 | 4.00 |
n(O2)/mol | 2.00 | 1.25 | 1.00 | n3 |
A. 5s��NO2��ƽ����Ӧ����Ϊ0.3 mol/(Ls��
B. ��10 sʱ�����������г���2 mol N2O5(g)������ƽ���£�NO2���������������
C. ��5 s ʱ�����ھ��Ⱥ����´�ƽ�⣬��ƽ���µ�ƽ�ⳣ����ԭƽ���С
D. ToC���÷�Ӧ��ƽ�ⳣ��Ϊ0.125����Ӧ���ƽ��ת���ʾ�Ϊ50%