��Ŀ����
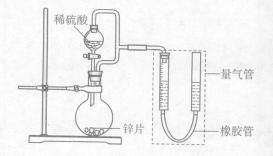
ijͬѧ���������ͼ��ʾװ�ã����ּг�װ������ȥ������ʵ���о���
��ش�
��1��������װ��̽��Ӱ�컯ѧ��Ӧ���ʵ����ء�
��Բ����ƿ�з�����Ӧ�����ӷ���ʽ�� ��
��������װ�ý���ʵ�飬������9��0 mL����Ϊ��ʱ�յ㣬���Ϊ ��
��
�Ƚ�ʵ��I�͢���Եó���ʵ������� ��
ʵ������У��������ܵIJ����� ��
������пƬ���ɺ����ʵĴ�пƬ���ҿ�����������ʹ��������ʵ����ȫһ�£�����õķ�Ӧ���ʾ���������ʵ���Ӧ�����ݡ���пƬ���������ʿ����ǣ�����ţ� ��
A��ʯī b���� c��ͭd��ɳ�����������裩
��2��������װ����֤�����ڳ�ʪ�����лᷢ��������ʴ��
��Բ����ƿ�е��Լ���ѡ�ã�����ţ� ��
a��NaOH��Һ b��C2H5OH c��NaCl��Һ d��ϡ����
����֤�������ڳ�ʪ�����лᷢ��������ʴ�������� ��

��ش�
��1��������װ��̽��Ӱ�컯ѧ��Ӧ���ʵ����ء�
��Բ����ƿ�з�����Ӧ�����ӷ���ʽ�� ��
��������װ�ý���ʵ�飬������9��0 mL����Ϊ��ʱ�յ㣬���Ϊ
 ��
��| ��� | V��H2SO4��/mL | C(H2SO4)/mol��L-1 | t/s |
| I | 40 | 1 | t1 |
| II | 40 | 4 | t2 |
ʵ������У��������ܵIJ����� ��
������пƬ���ɺ����ʵĴ�пƬ���ҿ�����������ʹ��������ʵ����ȫһ�£�����õķ�Ӧ���ʾ���������ʵ���Ӧ�����ݡ���пƬ���������ʿ����ǣ�����ţ� ��
A��ʯī b���� c��ͭd��ɳ�����������裩
��2��������װ����֤�����ڳ�ʪ�����лᷢ��������ʴ��
��Բ����ƿ�е��Լ���ѡ�ã�����ţ� ��
a��NaOH��Һ b��C2H5OH c��NaCl��Һ d��ϡ����
����֤�������ڳ�ʪ�����лᷢ��������ʴ�������� ��

��

��ϰ��ϵ�д�
�����Ŀ
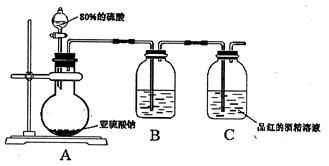




 �����ӵ�±ˮΪ��Ҫԭ���Ʊ���ˮ
�����ӵ�±ˮΪ��Ҫԭ���Ʊ���ˮ ��
�� ���������£�
���������£�
 ��
�� ���ɱ������ݿ�֪�������Ͽ�ѡ��pH���Χ�� ���ữ��ҺZʱ��ʹ�õ��Լ�Ϊ ��
���ɱ������ݿ�֪�������Ͽ�ѡ��pH���Χ�� ���ữ��ҺZʱ��ʹ�õ��Լ�Ϊ ��
 ���壬����װ���к������� ��
���壬����װ���к������� ��
 �ĵ��볣��
�ĵ��볣�� ��
�� ��
�� �ĵ�
�ĵ� �볣��
�볣�� ��
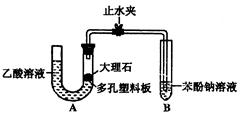
�� ��ijͬѧ���ʵ����֤
��ijͬѧ���ʵ����֤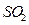 ��
�� ����ǰ�ߵ�
����ǰ�ߵ� ��
�� ��
�� ��
�� ������ˮ������ʯ��ˮ������
������ˮ������ʯ��ˮ������ .
.