��Ŀ����
4���ݱ�������ѧ��ͨ���Ե��ǽ��п����Է����Ƚ⣬������ȡһ�ֽ������������ṹ��������ij����Բ��ϣ����������ֲ��ϲ���������У������Ʊ�������ǿ�ȵĸ����ܻ����������й��ڶ��������˵���У���ȷ���ǣ�������| A�� | ������������ˮ�����ԣ����Զ��������������������� | |
| B�� | ��������̼ͨ���������Һ���Եõ����� | |
| C�� | ��Ϊ����ʱ����������̼���Ʒ�Ӧ�ų�������̼�����Թ�������Ա�̼��ǿ | |
| D�� | ������������������������������ |
���� A���������費����ˮ��
B��̼�������ǿ�ڹ��
C���Ƚ�����ǿ����Ӧ����Һ�н��У�
D����������������ᷴӦ�����ķ������ˮ��
��� �⣺A���������費����ˮ����A����
B��̼�������ǿ�ڹ��ᣬ������̼ͨ���������Һ���Եõ����ᣬ��B��ȷ��
C���Ƚ�����ǿ����Ӧ����Һ�н��У��ڸ��������·�Ӧ����Ҫ�������ε��ȶ��ԣ���C����
D����������������ᷴӦ�����ķ������ˮ���ܹ�������ḯʴ����D����
��ѡ��B��
���� ���⿼���˹輰�仯��������ʣ���Ϥ��������������ǽ���ؼ���������Ԫ�ػ�����֪ʶ���ۺ���������õĿ��飬�ѶȲ���ע����ػ���֪ʶ�Ļ��ۣ�

��ϰ��ϵ�д�
�����Ŀ
14������ָ�������µĽ����ȷ���ǣ�������
| A�� | 0.1mol•L?1��ˮ�У�c��OH-��=c��NH4+�� | |
| B�� | 10mL 0.02mol•L?1HCl��Һ��10mL 0.02mol•L?1 Ba��OH��2��Һ��ֻ�ϣ�����Ϻ���Һ�����Ϊ20mL������Һ��pH=7 | |
| C�� | ��0.1mol•L?1 CH3COONa��Һ�У�c��OH?��=c��CH3COOH��+c��H+�� | |
| D�� | 0.1mol•L?1ij��Ԫ������NaHA��Һ�У�c��Na+��=2c��A2?��+c��HA?��+c��H2A�� |
15�����ǵ���ʳס���ж��̺��ŷḻ�Ļ�ѧ֪ʶ������˵������ȷ���ǣ�������
| A�� | �����еIJ�˿�͵��ڶ����ںϳ��л��߷��Ӳ��� | |
| B�� | �ӵ�ʳ�κ���ǿ�����Ͷ����Բ�������������Ԫ�� | |
| C�� | ������¥����ʱ�õ��ĸ��Ǵ����� | |
| D�� | ��Һ��ʯ������������������ȼ�ϣ�������ɿ�����Ⱦ |
12������ѡ���У���Ӧ����Һ�ĵ��������ȷ�Ӧǰ������ǿ���ǣ�������
| A�� | ��100mL 1mol/L������Һ�м���4g NaOH���� | |
| B�� | ��100mL 1mol/L�Ĵ�����Һ��ͨ����NH3���� | |
| C�� | �ͺͳ���ʯ��ˮ��ͨ����CO2���� | |
| D�� | ͨ����������������������Һ�� |
19����NA��ʾ�����ӵ�������ֵ��������������ȷ���ǣ�������
| A�� | ���³�ѹ�£�6gO2��26gO3��������е�ԭ������Ϊ2NA | |
| B�� | 100mL 0.1mol/L��NaCl��Һ�У�����������Ϊ0.02NA | |
| C�� | 23g�����Ʊ�Ϊ������ʱ�õ��ĵ�����ΪNA | |
| D�� | ��״���£�2.24LH2O������ԭ����Ϊ0.3NA |
5���й�����˵����ȷ���ǣ�������
| A�� | �ø��������Һ�ζ�Na2SO3��Һ���յ㣬�������һ�θ��������Һ����Һ��ɫǡ�����Ϻ�ɫ��Ϊ��ɫ�Ұ���Ӳ���ɫ | |
| B�� | ȼ�ϵ��������ʵ���У�ʯī���ú��������Ϊ�˸��õĹ̶��缫 | |
| C�� | pH��ֽ�ڼ����Һʱ����ʪ��ʹ��ʯ����ֽ�������ʱ��Ҫʪ��ճ�ڽྻ��������һ�� | |
| D�� | ��������ȡ�⣬�ɽ��һ���ĺ�����ˮ���һ��ʱ�����ˣ�����Һ�м���ϡ�����ת�Ƶ���Һ©������CCl4��ȡ���� |
6���������ʹ���������������õ���Ҫ�о�����
Ti��BH4��3��һ�ִ�����ϣ�����TiCl4��LiBH4��Ӧ�Ƶã�
��1����̬Ti3+��δ�ɶԵ�������1����
��2��LiBH4��Li+��BH4-���ɣ�LiBH4�в����ڵ���������C�����ţ���
A�����Ӽ� B�����ۼ� C�������� D����λ��
��3��Li��B��HԪ�صĵ縺���ɴ�С����˳��ΪH��B��Li��
��4������X����ͨ������γɡ���״�ṹ������ΪDZ�ڵĴ�����ϣ�Xһ������BC�����ţ���
A��H2O B��CH4 C��HF D��CO��NH2��2
��5���ء�þ�����γɵ�ij������ľ���ṹΪK+���������������ģ�Mg2+�ھ�����8�����ǣ�F-���ھ�����������ģ��ɼء�þ�����γɵĸû�����Ļ�ѧʽΪKMgF3��ÿ��K+��12��F-��λ��
��6���жϺ���������ǿ����һ����������Ǻ�������ӽṹ�к����ǻ���ԭ����Խ�࣬�ú����������Խǿ�����±���ʾ������������ǿ������ǻ���ԭ�����Ĺ�ϵ
������H3PO3��������H3AsO3����ʽ���ƣ������ǵ����Բ��ܴ�H3PO3����ǿ�ᣬH3AsO3�������������������ԣ��ɴ˿��Ƴ�H3AsO3�ṹʽΪ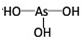 ��H3PO3��H3AsO3�������NaOH��Һ��Ӧ�Ļ�ѧ����ʽ�ֱ��ǣ�
��H3PO3��H3AsO3�������NaOH��Һ��Ӧ�Ļ�ѧ����ʽ�ֱ��ǣ�
��H3PO3+2NaOH=Na2HPO3+2H2O��
��H3AsO3+3NaOH=Na3AsO3+3H2O��
Ti��BH4��3��һ�ִ�����ϣ�����TiCl4��LiBH4��Ӧ�Ƶã�
��1����̬Ti3+��δ�ɶԵ�������1����
��2��LiBH4��Li+��BH4-���ɣ�LiBH4�в����ڵ���������C�����ţ���
A�����Ӽ� B�����ۼ� C�������� D����λ��
��3��Li��B��HԪ�صĵ縺���ɴ�С����˳��ΪH��B��Li��
��4������X����ͨ������γɡ���״�ṹ������ΪDZ�ڵĴ�����ϣ�Xһ������BC�����ţ���
A��H2O B��CH4 C��HF D��CO��NH2��2
��5���ء�þ�����γɵ�ij������ľ���ṹΪK+���������������ģ�Mg2+�ھ�����8�����ǣ�F-���ھ�����������ģ��ɼء�þ�����γɵĸû�����Ļ�ѧʽΪKMgF3��ÿ��K+��12��F-��λ��
��6���жϺ���������ǿ����һ����������Ǻ�������ӽṹ�к����ǻ���ԭ����Խ�࣬�ú����������Խǿ�����±���ʾ������������ǿ������ǻ���ԭ�����Ĺ�ϵ
| ������ | ���� | ���� | ������ | |
| ������ | Cl-OH | 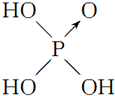 |  | 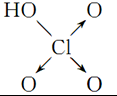 |
| ���ǻ���ԭ���� | 0 | 1 | 2 | 3 |
| ���� | ���� | ��ǿ�� | ǿ�� | ��ǿ�� |
 ��H3PO3��H3AsO3�������NaOH��Һ��Ӧ�Ļ�ѧ����ʽ�ֱ��ǣ�
��H3PO3��H3AsO3�������NaOH��Һ��Ӧ�Ļ�ѧ����ʽ�ֱ��ǣ���H3PO3+2NaOH=Na2HPO3+2H2O��
��H3AsO3+3NaOH=Na3AsO3+3H2O��
