��Ŀ����
4�����й�����Һ�����ӵ�˵����ȷ���ǣ�������| A�� | 0.1 mol•L-1��Na2CO3��Һ������Ũ�ȹ�ϵ��c ��Na+��=2c ��CO32-��+c ��HCO3-��+c ��H2CO3�� | |
| B�� | 0.1 mol•L-1��NH4Cl��0.1 mol•L-1��NH3•H2O�������Ϻ���Һ�е�����Ũ�ȹ�ϵ��c ��Cl-����c ��NH4+����c ��H+����c ��OH-�� | |
| C�� | �����£���������Һ�еμ���������ʹ��Һ��pH=7��������Һ�У�����Ũ�ȹ�ϵ��c ��Na+����c ��CH3COO-�� | |
| D�� | 0.1 mol•L-1��NaHS��Һ������Ũ�ȹ�ϵ��c ��OH-��=c ��H+��-c ��S2-��+c ��H2S�� |
���� A������̼������Һ�е������غ�����жϣ�
B��Ũ�����ʱ��һˮ�ϰ�����̶ȴ���笠����ӵ�ˮ��̶ȣ����Һ�ʼ��ԣ���c��OH-����c��H+�������ݵ���غ��֪��c��NH4+����c��Cl-����
C����Һ��pH=7����c��OH-��=c��H+�������ݵ���غ��֪c ��Na+��=c ��CH3COO-����
D�����ݻ��Һ�еĵ���غ�������غ�����жϣ�
��� �⣺A�����������غ��֪��c��Na+��=2c��CO32-��+2c��HCO3-��+2c��H2CO3������A����
B.0.1 mol•L-1��NH4Cl��0.1 mol•L-1��NH3•H2O�������Ϻ�һˮ�ϰ�����̶ȴ���笠����ӵ�ˮ��̶ȣ����Һ�ʼ��ԣ���c��OH-����c��H+�������ݵ���غ��֪��c��NH4+����c��Cl-���������Һ������Ũ�ȴ�СΪ��c��NH4+����c��Cl-����c��OH-����c��H+������B����
C�������£���������Һ�еμ���������ʹ��Һ��pH=7����c��OH-��=c��H+�������ݵ���غ��֪c ��Na+��=c ��CH3COO-������C����
D����Һ�д��ڵ���غ㣺c��Na+��+c��H+��=c��HS-��+2c��S2-��+c��OH-�������������غ�ã�c��Na+��=c��H2S��+c��HS-��+c��S2-�������Ե�c��S2-��+c��OH-��=c��H+��+c��H2S������c ��OH-��=c��H+��-c��S2-��+c��H2S������D��ȷ��
��ѡD��
���� ���⿼��������Ũ�ȴ�С�ıȽϣ���Ŀ�Ѷ��еȣ���ȷ��Һ�е����ʼ��������ǽⱾ��ؼ�����ϵ���غ�������غ��������������������ѧ���ķ������������Ӧ��������

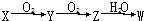 ��֪X��Y��Z��W����ͬһԪ�أ�����ͼ��ʾ��ת����ϵ����X��һ�ֵ��ʣ�WΪһ�ֺ����ᣬ��X�������ǣ�������
��֪X��Y��Z��W����ͬһԪ�أ�����ͼ��ʾ��ת����ϵ����X��һ�ֵ��ʣ�WΪһ�ֺ����ᣬ��X�������ǣ�������| A�� | N2 | B�� | C | C�� | S | D�� | Si |
| A�� | ������ˮ | |
| B�� | ���ɵ����������ڷŵ������·�Ӧ���� | |
| C�� | ������������������ | |
| D�� | ����ɫ���� |
| A�� | ���������������������ֱ���ȫȼ�գ����߷ų��������� | |
| B�� | ��C�����ʯ����C��ʯī������H=-1.9KJ/mol ��֪�����ʯ��ʯī�ȶ� | |
| C�� | ��101Kpaʱ��2gH2��ȫȼ������Һ̬ˮ���ų�285.8KJ����������ȼ�յ��Ȼ�ѧ����ʽΪ��2H2��g��+O2��g��=2H2O��l������H=+285.8KJ/mol | |
| D�� | ϡ��Һ�У�H+��aq��+OH-��aq��=H2O��l������H=-53.7KJ/mol��������0.5 molH2SO4��Ũ��Һ�뺬1 molNaOH����Һ��ϣ��ų�����������53.7KJ/mol |
| A�� | �÷�ɢϵ�ķ�ɢ��ΪFe2O3 | |
| B�� | ���ù��˵ķ�������ɫ������������Na+���뿪 | |
| C�� | �ڵ糡�����£�����������ɢϵ��ɫ�����˵���÷�ɢϵ������� | |
| D�� | ����NaOHʱ�����ķ�Ӧ����Ϊ��Fe2++2Fe3++8OH-�TFe3O4+4H2O |
| A�� | 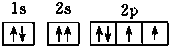 | B�� | 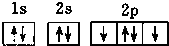 | ||
| C�� | 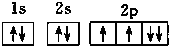 | D�� |  |