��Ŀ����
�±���Ԫ�����ڱ���һ���֣��ش������й����⣺
��1����-��Ԫ���У�ijԪ�ص�������ȿ���Ư�����ǿ��������ձ��ļ�����Ԫ���� ����Ԫ�����ƣ��������������еĻ�ѧ������Ϊ ������Լ����Ǽ��Լ�������
��2������ЩԪ�ص�����������ˮ�����У�������ǿ���� ���ѧʽ���������Ե����������� ���ѧʽ����д�����߷�Ӧ�����ӷ���ʽ ��
��3��д����ҵ��Ԫ�آ���⻯����������Ļ�ѧ����ʽ ��
��4���õ���ʽ��ʾԪ�آں�Ԫ�آ���ɵĻ�������γɹ��� ��
��5��Ԫ�آ���Ԫ�آ��γɵĻ��������ܽ�Ԫ�آĵ��ʣ��û�����ĵ���ʽΪ ��
��6����Ԫ�آ���Ԫ�آ��У���ѧ���ʽϻ��õ��� ����Ԫ�ط��ţ���֤���ý��۵Ļ�ѧʵ�鼰�������� ��
| ���� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| 2 | �� | �� | �� | |||||
| 3 | �� | �� | �� | �� | �� | |||
| 4 | �� | �� |
��2������ЩԪ�ص�����������ˮ�����У�������ǿ����
��3��д����ҵ��Ԫ�آ���⻯����������Ļ�ѧ����ʽ
��4���õ���ʽ��ʾԪ�آں�Ԫ�آ���ɵĻ�������γɹ���
��5��Ԫ�آ���Ԫ�آ��γɵĻ��������ܽ�Ԫ�آĵ��ʣ��û�����ĵ���ʽΪ
��6����Ԫ�آ���Ԫ�آ��У���ѧ���ʽϻ��õ���
���㣺Ԫ�������ɺ�Ԫ�����ڱ����ۺ�Ӧ��
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
��������Ԫ�������ڱ���λ�ã�֪��ΪC����ΪF����ΪNa����ΪMg����ΪAl����ΪP����ΪCl����Ϊk����ΪCa����ΪN��
��1������Ԫ���У�ijԪ�ص�������ȿ���Ư�����ǿ��������ձ��ļ�����������Ϊ��������ͬԪ��֮���γɵĻ�ѧ��Ϊ���Լ���
��2������Ԫ��������������ˮ�����У�������ǿ�Ǹ����ᣬ������������������������߷����кͷ�Ӧ������Ӧ������ˮ��
��3��������������������NO��ˮ��
��4��Ԫ�آں�Ԫ�آ���ɵĻ�����ΪCaF2���ɸ�����������ӹ��ɣ���Caԭ�ӡ�Fԭ�ӵ���ʽ��ʾ���γɹ��̣�
��5��Ԫ�آ���Ԫ�آ��γɵĻ��������ܽ�Ԫ�آĵ��ʣ��û�����ΪCS2���ṹԽ������̼���ƣ�������Cԭ����Sԭ��֮���γ�2�Թ��õ��Ӷԣ�
��6��ͬ����������ҽ����Լ�������ѧ���ʻ����Լ������������ý���������ˮ�ķ�Ӧ������֤��
��1������Ԫ���У�ijԪ�ص�������ȿ���Ư�����ǿ��������ձ��ļ�����������Ϊ��������ͬԪ��֮���γɵĻ�ѧ��Ϊ���Լ���
��2������Ԫ��������������ˮ�����У�������ǿ�Ǹ����ᣬ������������������������߷����кͷ�Ӧ������Ӧ������ˮ��
��3��������������������NO��ˮ��
��4��Ԫ�آں�Ԫ�آ���ɵĻ�����ΪCaF2���ɸ�����������ӹ��ɣ���Caԭ�ӡ�Fԭ�ӵ���ʽ��ʾ���γɹ��̣�
��5��Ԫ�آ���Ԫ�آ��γɵĻ��������ܽ�Ԫ�آĵ��ʣ��û�����ΪCS2���ṹԽ������̼���ƣ�������Cԭ����Sԭ��֮���γ�2�Թ��õ��Ӷԣ�
��6��ͬ����������ҽ����Լ�������ѧ���ʻ����Լ������������ý���������ˮ�ķ�Ӧ������֤��
���
�⣺��Ԫ�������ڱ���λ�ã�֪��ΪC����ΪF����ΪNa����ΪMg����ΪAl����ΪP����ΪCl����Ϊk����ΪCa����ΪN��
��1������Ԫ���У�ijԪ�ص�������ȿ���Ư�����ǿ��������ձ��ļ�����������Ϊ��������Ӧ��Ԫ��ΪS������������S��OԪ��֮���γɵĻ�ѧ��Ϊ���Լ����ʴ�Ϊ�����Լ���
��2������Ԫ��������������ˮ�����У�������ǿ��HClO4��Al��OH��3����������������߷����кͷ�Ӧ������Ӧ������ˮ����Ӧ���ӷ���ʽΪ��Al��OH��3+3H+�TAl3++3H2O��
�ʴ�Ϊ��HClO4��Al��OH��3��Al��OH��3+3H+�TAl3++3H2O��
��3��������������������NO��ˮ����Ӧ����ʽΪ��4NH3+5O2
4NO+6H2O���ʴ�Ϊ��4NH3+5O2
4NO+6H2O��
��4��Ԫ�آں�Ԫ�آ���ɵĻ�����ΪCaF2���ɸ�����������ӹ��ɣ���Caԭ�ӡ�Fԭ�ӵ���ʽ��ʾ���γɹ���Ϊ�� ��
��
�ʴ�Ϊ�� ��
��
��5��Ԫ�آ���Ԫ�آ��γɵĻ��������ܽ�Ԫ�آĵ��ʣ��û�����ΪCS2���ṹԽ������̼���ƣ�������Cԭ����Sԭ��֮���γ�2�Թ��õ��Ӷԣ������ʽΪ��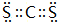 ���ʴ�Ϊ��
���ʴ�Ϊ��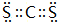 ��
��
��6��ͬ����������ҽ����Լ������ʻ�����Na��Mg��ʵ�鷽��������Ϊ��������ˮ���ҷ�Ӧ��þ����ˮ������Ӧ���ʴ�Ϊ��Na��������ˮ���ҷ�Ӧ��þ����ˮ������Ӧ��
��1������Ԫ���У�ijԪ�ص�������ȿ���Ư�����ǿ��������ձ��ļ�����������Ϊ��������Ӧ��Ԫ��ΪS������������S��OԪ��֮���γɵĻ�ѧ��Ϊ���Լ����ʴ�Ϊ�����Լ���
��2������Ԫ��������������ˮ�����У�������ǿ��HClO4��Al��OH��3����������������߷����кͷ�Ӧ������Ӧ������ˮ����Ӧ���ӷ���ʽΪ��Al��OH��3+3H+�TAl3++3H2O��
�ʴ�Ϊ��HClO4��Al��OH��3��Al��OH��3+3H+�TAl3++3H2O��
��3��������������������NO��ˮ����Ӧ����ʽΪ��4NH3+5O2
| ||
| �� |
| ||
| �� |
��4��Ԫ�آں�Ԫ�آ���ɵĻ�����ΪCaF2���ɸ�����������ӹ��ɣ���Caԭ�ӡ�Fԭ�ӵ���ʽ��ʾ���γɹ���Ϊ��
 ��
���ʴ�Ϊ��
 ��
����5��Ԫ�آ���Ԫ�آ��γɵĻ��������ܽ�Ԫ�آĵ��ʣ��û�����ΪCS2���ṹԽ������̼���ƣ�������Cԭ����Sԭ��֮���γ�2�Թ��õ��Ӷԣ������ʽΪ��
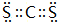 ���ʴ�Ϊ��
���ʴ�Ϊ��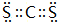 ��
����6��ͬ����������ҽ����Լ������ʻ�����Na��Mg��ʵ�鷽��������Ϊ��������ˮ���ҷ�Ӧ��þ����ˮ������Ӧ���ʴ�Ϊ��Na��������ˮ���ҷ�Ӧ��þ����ˮ������Ӧ��
���������⿼��Ԫ�����ڱ���Ԫ�������ɣ��ѶȲ���ע���Ԫ�����ڱ���������գ������õ���ʽ��ʾ��ѧ�������ʵ��γɣ�

��ϰ��ϵ�д�
�����Ŀ
��NA��ʾ����٤������������˵������ȷ���ǣ�������
| A��1mol 12C18O2�У�������������Ϊ22NA |
| B��5.6g�������ᷴӦʧȥ�ĵ�����һ��Ϊ0.2NA |
| C�������£�42g C2H4��C4H8�Ļ�����к��е�̼ԭ����Ϊ3NA |
| D������£�11.2L SO3�����ķ�����Ϊ0.5NA |
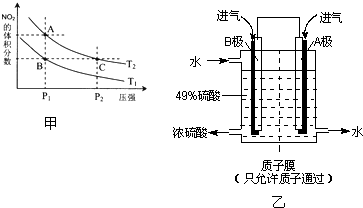
 ͼ��X��Y��Z��W�ǵؿǺ���ǰ��λ������˳��Ԫ����ɵĵ��ʣ�����Ϊ�����A�д��ԣ�����֮���������ת����ϵ�����ֲ�������ȥ����
ͼ��X��Y��Z��W�ǵؿǺ���ǰ��λ������˳��Ԫ����ɵĵ��ʣ�����Ϊ�����A�д��ԣ�����֮���������ת����ϵ�����ֲ�������ȥ����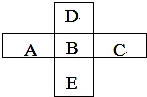 A��B��C��D��E��������Ԫ���������ڱ���λ����ͼ��ʾ��
A��B��C��D��E��������Ԫ���������ڱ���λ����ͼ��ʾ��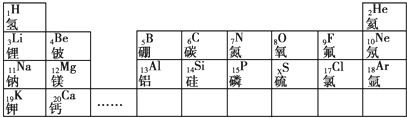
 ��ʾ���ǣ�д���ӷ��ţ�
��ʾ���ǣ�д���ӷ��ţ�