��Ŀ����
9������˵����ȷ���ǣ�������| A�� | 1 Lˮ���ܽ�1 mol NaCl���γ���Һ�����ʵ���Ũ��Ϊ1 mol•L-1 | |
| B�� | ��״���£�22.4 L HCl����1 Lˮ���γɵ���Һ���Ϊ1 L | |
| C�� | ��״���£���33.6 L HCl����ˮ�γ�1 L��Һ�������ʵ���Ũ����1.5 mol•L-1 | |
| D�� | 1 mol CaCl2����ˮ�γ�1 L��Һ��������Һ��c��Cl-������1 mol•L-1 |
���� A�����ʵ���Ũ���У����Ϊ��Һ����������ܼ��������
B����״����22.4L HCl����1Lˮ��������Һ�����������1L��
C.33.6LHCl�����ʵ���Ϊ1.5mol���ݴ˼��㼴�ɣ�
D.1molCaCl2�к���2mol�����ӣ�
��� �⣺A.1 Lˮ���ܽ�1 mol NaCl���γ���Һ���������1L����Ũ��С��1mol/L����A����
B�����ʵ���Ũ���У����Ϊ��Һ����������ܼ����������B����
C.33.6 L HCl����ˮ�γ�1 L��Һ���õ������ʵ�����1.5mol����Һ�����Ϊ1L����Ũ��Ϊ1mol/L����C��ȷ��
D.1 mol CaCl2����ˮ�γ�1 L��Һ��������Һ��c��Cl-������2mol•L-1����D����
��ѡC��
���� ���⿼��һ�����ʵ���Ũ����Һ�����ƣ��ѶȲ�����ȷ���ʵ���Ũ�ȶ����ǽ���ؼ���ע�����Ϊ��Һ�������

��ϰ��ϵ�д�
�����Ŀ
19����һ���¶��£����ܱ������м���I2��H2��1mol��������Ӧ��H2��g��+I2��g��?2HI��g������ƽ��ʱ������1.6mol HI���������������䣬��ʼ�����H2��Ϊ1.2mol����ﵽƽ��ʱ������HI���ʵ�����������
| A�� | 1.4mol | B�� | 1.6mol | C�� | 1.8mol | D�� | 2.0mol |
20��ijԪ��M2+���������Ϊ24����Ԫ��������ԭ���еģ�������
| A�� | ${\;}_{24}^{52}$Cr | B�� | ${\;}_{12}^{24}$Mg | C�� | ${\;}_{26}^{56}$Fe | D�� | ${\;}_{22}^{48}$Ti |
17��һ������ȼ�ϵ�أ����Զ������Ϊ�缫����KOH��Һ�У�Ȼ��ֱ���������ͨ�������������缫��ӦʽΪ��C2H6+18OH--14e-�T2CO32-+12H2O��7H2O+$\frac{7}{2}$O2+14e-�T14OH-���йش˵�ص��ƶ���ȷ���ǣ�������
| A�� | �������Һ�е����������ƶ� | |
| B�� | �ŵ�һ��ʱ���KOH�����ʵ���Ũ�Ȳ��� | |
| C�� | ͨ����ĵ缫Ϊ���� | |
| D�� | �μӷ�Ӧ��O2��C2H6�����ʵ���֮��Ϊ2��7 |
4������ʵ����Ƽ����Ӧ�����ӷ���ʽ����ȷ���ǣ�������
| A�� | ��NaAlO2��Һ��ͨ�����CO2��2AlO2-+CO2+3H2O�T2Al��OH��3��+CO32- | |
| B�� | ����SO2ͨ�뱽������Һ�У�C6H5O-+SO2+H2O�TC6H5OH+HSO3- | |
| C�� | ���ҽ���Һ�м���ϡ���ᡢ˫��ˮ��2I-+2H++H2O2�TI2+2H2O | |
| D�� | ��Ũ�����ữ��KMnO4��Һ��H2O2��Ӧ��֤��H2O2���л�ԭ�ԣ�2MnO4-+6H++5H2O2�T2Mn2++5O2��+8H2O |
14��ָ�����£�1mol��ѧ���ֽ����̬ԭ������Ҫ��������E��ʾ������±���Ϣ�жϲ���ȷ���ǣ�������
| ���ۼ� | H-H | F-F | H-F | H-Cl | H-I |
| E��kJ•mol -1 �� | 436 | 157 | 568 | 432 | 298[ |
| A�� | H2��g��+F2��g���T2HF��g����H=-25 kJ•mol -1 | |
| B�� | �������ȶ��Ĺ��ۼ���H-F�� | |
| C�� | H 2��g����2H��g����H=+436 kJ•mol -1 | |
| D�� | 432 kJ•mol -1��E��H-Br����298 kJ•mol -1 |
1�����ΪV mL���ܶ�Ϊd g/cm3����Һ��������Է�������ΪM������m g�������ʵ���Ũ��Ϊcmol/L����������Ϊ��%�����б���ʽ����ȷ���ǣ�������
| A�� | c=$\frac{1000��d}{M}$ | B�� | m=Vd | C�� | m=Vd�� | D�� | c=$\frac{1000m}{VM}$m |
18��ijѧ���������ͼ�ķ�����A�ν��м������ɴ˷��������н����У���ȷ���ǣ�������
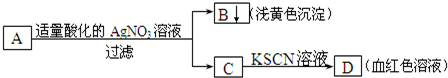

| A�� | A��һ����Fe2+ | B�� | BΪAgI���� | ||
| C�� | A�п�����Fe3+ | D�� | Aһ��ΪFeBr2��Һ |