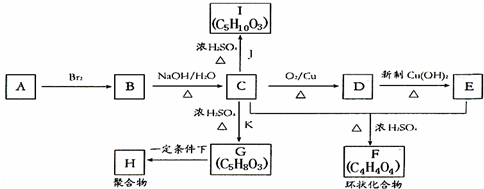
��Ŀ����
4��PETG��һ�����Ͳ��ϣ��ɻ������ã��Ի����������κ���в����ṹ��ʽ���£�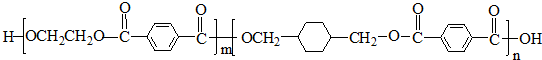
��֪��RCOOR1+R2OH-��RCOOR2+R1OH��R��R1��R2��ʾ��������������ͼ��ʾ�ĺϳ�·�߿ɺϳ�PETG��
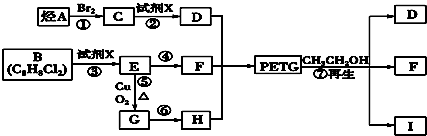
�Իش��������⣺
��1������������Ӧ�У�����ȡ����Ӧ���Тڢۢߣ���д��ţ���
��2��д���ṹ��ʽ��B
 ��I
��I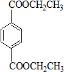 ��
����3��д����ѧ����ʽ��
��Ӧ��

��Ӧ��
 ��
����4���ϳ�ʱӦ���Ƶĵ�������ʵ���n��D����n��F����n��H��=m��n����m+n������m��n��ʾ����
���� ��PETG�Ľṹ��ʽ��֪��Ӧ�ĵ�����HOCH2CH2OH��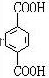 ��
�� ����DӦΪHOCH2CH2OH��FΪ
����DӦΪHOCH2CH2OH��FΪ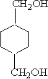 ��HΪ
��HΪ ��EΪ
��EΪ ��GΪ
��GΪ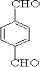 ��BΪ
��BΪ ��AΪCH2=CH2��CΪCH2BrCH2Br��IΪ
��AΪCH2=CH2��CΪCH2BrCH2Br��IΪ ������л���Ľṹ�������Լ���ĿҪ��ɽ����⣮
������л���Ľṹ�������Լ���ĿҪ��ɽ����⣮
��� �⣺��PETG�Ľṹ��ʽ��֪��Ӧ�ĵ�����HOCH2CH2OH�� ��
�� ����DӦΪHOCH2CH2OH��FΪ
����DӦΪHOCH2CH2OH��FΪ ��HΪ
��HΪ ��EΪ
��EΪ ��GΪ
��GΪ ��BΪ
��BΪ ��AΪCH2=CH2��CΪCH2BrCH2Br��IΪ
��AΪCH2=CH2��CΪCH2BrCH2Br��IΪ ��
��
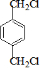
��1�������Ϸ���������ת����ϵ��֪�ڢۢ�����ȡ����Ӧ���٢�Ϊ�ӳɷ�Ӧ������Ϊ������Ӧ��
�ʴ�Ϊ���ڢۢߣ�
��2�������Ϸ�����֪BΪ ��IΪ
��IΪ ��
��
�ʴ�Ϊ�� ��
�� ��
��
��3����Ӧ�ܵķ�ӦΪ ����Ӧ�ݵķ�ӦΪ
����Ӧ�ݵķ�ӦΪ ��
��
�ʴ�Ϊ�� ��
�� ��
��
��4���ϳ�ʱӦ���Ƶĵ�������ʵ���n ��D����n ��F����n ��H��=m��n����m+n����
�ʴ�Ϊ��m��n����m+n����
���� ���⿼���л�����ƶϣ�������ѧ�������������ƶ������Ŀ��飬������Ϣ����ע����ݸ߾���Ľṹ��ʽ�жϵ���Ϊ�������ͻ�ƿڣ�����ʱע����������Ϣ��Ϊ������Ĺؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ����ν����Ž̲��㽭���̴�ѧ������ϵ�д�
����ν����Ž̲��㽭���̴�ѧ������ϵ�д� �����Ļ������������������ϵ�д�
�����Ļ������������������ϵ�д�
�����Ŀ
1��NA��ʾ�����ӵ�������ֵ������˵����ȷ���ǣ�������
| A�� | ���³�ѹ�£�1.6gCH4���еĵ�����ĿΪNA | |
| B�� | ��״���£�2.2 LCCl4���е�̼ԭ����ĿΪ0.1NA | |
| C�� | 101kPa��0��ʱ��22.4LH2����ԭ����ΪNA�� | |
| D�� | 1mol/L ��NaOH��Һ�У�Na+����ĿΪNA |
12������ʽΪC4H8�Һ�һ��֧������״�л���Ķ��ȴ�������У�������˳���칹����������
| A�� | 4�� | B�� | 5�� | C�� | 6�� | D�� | 7�� |
19����22.4g������ȫ�ܽ���ijŨ�ȵ������У��練Ӧֻ�ռ���0.3molNO2��0.2molNO������˵����ȷ���ǣ�������
| A�� | ��Ӧ�����ɵ���ֻ��Fe��NO3��3 | |
| B�� | ��Ӧ�����ɵ���ֻ��Fe��NO3��2 | |
| C�� | ��Ӧ�����ɵ���ΪFe��NO3��3��Fe��NO3��2�������ʵ���֮��Ϊ3��1 | |
| D�� | ��Ӧ�����ɵ���ΪFe��NO3��3��Fe��NO3��2�������ʵ���֮��Ϊ1��3 |
9����ʵ����Ϊ�ⶨһ�����Ƶ�ϡ�����ȷŨ�ȣ�ͨ�����ô�����Na2CO3����ˮ����ɱ���Һ�ζ�����������ǣ���ȡwg��������ˮNa2CO3װ����ƿ�У�����������ˮ�ܽ⣬����ʽ�ζ����м����������ζ�����1����ƿ��Ӧ���뼸��ָʾ��������CO2�ܽ�����Һ�л�Ӱ��pH����ȷ�Կ��ǣ��ζ��յ���ѡ��pHΪ4��5֮�䣬��ôָʾ����ѡ�����ζ�����Һ�ɻ� ɫ��ɳ�ɫʱ������ʾ�����յ㣻
�����к͵ζ����ⶨ�ռ�Ĵ��ȣ����ռ��в��������ᷴӦ�����ʣ��Ը���ʵ��ش�
��1��ȷ��ȡ�ռ���Ʒ5.0g������Ʒ���250mL�Ĵ���Һ��
��2��ȡ10.00mL����Һ���ü�ʽ�ζ�����ȡע����ƿ�У�����������
��3����0.2000mol/L��������Һ�ζ������ռ���Һ���ζ�ʱ������ת��ʽ�ζ��ܵIJ������������֣�����ע�ӣ�ֱ���ζ��յ㣮
��4���������вⶨ���ݣ������õ��������ݣ���������ռ���Һ��Ũ�ȣ���
��5�����������ⶨ���ݣ������õ��������ݣ������ռ�Ĵ��ȣ�
�����к͵ζ����ⶨ�ռ�Ĵ��ȣ����ռ��в��������ᷴӦ�����ʣ��Ը���ʵ��ش�
��1��ȷ��ȡ�ռ���Ʒ5.0g������Ʒ���250mL�Ĵ���Һ��
��2��ȡ10.00mL����Һ���ü�ʽ�ζ�����ȡע����ƿ�У�����������
��3����0.2000mol/L��������Һ�ζ������ռ���Һ���ζ�ʱ������ת��ʽ�ζ��ܵIJ������������֣�����ע�ӣ�ֱ���ζ��յ㣮
��4���������вⶨ���ݣ������õ��������ݣ���������ռ���Һ��Ũ�ȣ���
| �ζ����� | ����Һ���/mL | ���������/mL | |
| �ζ�ǰ������mL�� | �ζ��������mL�� | ||
| ��һ�� | 10.00 | 0.50 | 20.40 |
| �ڶ��� | 10.00 | 4.00 | 24.10 |
| ������ | 10.00 | 4.20 | 25.70 |
13��ϡ��Ԫ����ָԪ�����ڱ���ԭ������Ϊ57 ��71 ��15����ϵԪ�أ��Լ�����ϵԪ�ػ�ѧ�������Ƶ��֣�Sc�����ƣ�Y����17 ��Ԫ�أ���ش��������⣺
��1���֣�Sc��Ԫ�ص�ԭ�Ӻ�������Ų�ʽΪ1s22s22p63s23p63d14s2���أ�Ho���Ļ�̬ԭ�ӵ����Ų�ʽΪ[Xe]4f116s2��һ����̬��ԭ��������δ�ɶԵ�����Ϊ3��
��2��ϡ��Ԫ������Ļ��ϼ�Ϊ+3�ۣ���Ҳ����������+4�ۣ�������±��еĵ����������жϱ����������+4�۵�Ԫ����Ce��
����ϡ��Ԫ�صĵ����ܣ���λ��kJ•mol-1��
��3�����ӻ����� Na3[Sc��OH��6]�У����ڵ������������Ӽ���й��ۼ�����λ����
��4��Sm���̣��ĵ�����1��2-��������ɷ������·�Ӧ��Sm+ICH2CH2I��SmI2+CH2=CH2��
ICH2CH2I��̼ԭ���ӻ��������Ϊsp3��1mol CH2=CH2�к��еĦҼ���ĿΪ5NA��
��5��PrO2���������裩�ľ���ṹ��CaF2���ƣ�������Pr���裩ԭ��λ�����ĺͶ�����PrO2���������裩�ľ�������9����ԭ�ӣ�
��6��Ce���棩����Ϊ�����������壬�侧������Ϊa=516pm��������Ce���棩ԭ�ӵ���λ��Ϊ12����ʽ��ʾCe���棩���ʵ��ܶ�Ϊ$\frac{4��140}{6.02��1{0}^{23}����516��1{0}^{-10}��^{3}}$g•cm-3�����ؼ�����������
��1���֣�Sc��Ԫ�ص�ԭ�Ӻ�������Ų�ʽΪ1s22s22p63s23p63d14s2���أ�Ho���Ļ�̬ԭ�ӵ����Ų�ʽΪ[Xe]4f116s2��һ����̬��ԭ��������δ�ɶԵ�����Ϊ3��
��2��ϡ��Ԫ������Ļ��ϼ�Ϊ+3�ۣ���Ҳ����������+4�ۣ�������±��еĵ����������жϱ����������+4�۵�Ԫ����Ce��
����ϡ��Ԫ�صĵ����ܣ���λ��kJ•mol-1��
| Ԫ�� | I1 | I2 | I3 | I1+I2+I3 | I4 |
| Sc���֣� | 633 | 1235 | 2389 | 4257 | 7019 |
| Y���ƣ� | 616 | 1181 | 1980 | 3777 | 5963 |
| La���磩 | 538 | 1067 | 1850 | 3455 | 4819 |
| Ce���棩 | 527 | 1047 | 1949 | 3523 | 3547 |
��4��Sm���̣��ĵ�����1��2-��������ɷ������·�Ӧ��Sm+ICH2CH2I��SmI2+CH2=CH2��
ICH2CH2I��̼ԭ���ӻ��������Ϊsp3��1mol CH2=CH2�к��еĦҼ���ĿΪ5NA��
��5��PrO2���������裩�ľ���ṹ��CaF2���ƣ�������Pr���裩ԭ��λ�����ĺͶ�����PrO2���������裩�ľ�������9����ԭ�ӣ�
��6��Ce���棩����Ϊ�����������壬�侧������Ϊa=516pm��������Ce���棩ԭ�ӵ���λ��Ϊ12����ʽ��ʾCe���棩���ʵ��ܶ�Ϊ$\frac{4��140}{6.02��1{0}^{23}����516��1{0}^{-10}��^{3}}$g•cm-3�����ؼ�����������
14�����з�Ӧ�����ڼӳɷ�Ӧ���ǣ�������
| A�� | CH4+Cl2$\stackrel{��}{��}$CH3Cl+HCl | |
| B�� |  +Br2$\stackrel{����}{��}$ +Br2$\stackrel{����}{��}$ +H2O +H2O | |
| C�� | 2CH3CH2OH+O2$��_{��}^{Cu��Ag}$2CH3CHO+2H2O | |
| D�� | CH2�TCH2+HCl��CH3CH2Cl |