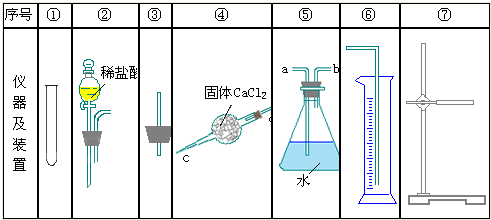
��Ŀ����
�ⶨ����ͭ���壨CuSO4?xH2O����xֵ��ʵ�������ͼ�����������գ�

��1����1�������______��
��2������ʱ����CuSO4?xH2O�������ɫ��______��ȫ��Ϊ______��ֹͣ���ȣ�
��3������Ҫ���к��ز�����ԭ����______��
��4�����γ������������±���
��CuSO4?xH2O�е�x=______��
��5�����в����ᵼ��xֵƫ�ߵ���______��ѡ���ţ�
a������δ����b�����ȹ������о��彦ʧ
c�����պ������ڿ�������ȴd�����ز���ʱ���������γ����Ľ��������0.01g��

��1����1�������______��
��2������ʱ����CuSO4?xH2O�������ɫ��______��ȫ��Ϊ______��ֹͣ���ȣ�
��3������Ҫ���к��ز�����ԭ����______��
��4�����γ������������±���
| ���� | ��1�� | ��2�� | ��3�� | ��4�� | ��5�� |
| ������g�� | m1 | m2 | m3 | m4 | m4 |
��5�����в����ᵼ��xֵƫ�ߵ���______��ѡ���ţ�
a������δ����b�����ȹ������о��彦ʧ
c�����պ������ڿ�������ȴd�����ز���ʱ���������γ����Ľ��������0.01g��
��1����������ͭ�����нᾧˮ�����������ȳ���������ͭ���������������ͼʾ���̣��ڶ��γ�����������������ͭ�������������һ�γ�����Ϊ����������
�ʴ�Ϊ��������
��2������ʱ����CuSO4?xH2O�������ɫ����ɫ��ɰ�ɫʱ������ͭ����ᾧˮ��ȫʧȥ��Ӧ��ֹͣ���ȣ�
�ʴ�Ϊ����ɫ����ɫ��
��3��ֻ������ͭ�����еĽᾧˮ��ȫʧȥ������ʹ�����������ȷ��������Ҫ���к��ز�����ʹ���γ�����������С��0.1g��
�ʴ�Ϊ��ȷ������ͭ������ȫʧȥ�ᾧˮ��
��4����ɫ����ͭ������Ϊ����m4-m1��g�����ʵ���Ϊ��
mol���ᾧˮ�����ʵ���Ϊ��
mol�����Խᾧˮx=
=
��
�ʴ�Ϊ��
��
��5��a������δ����൱������ͭ�����нᾧˮ�������ⶨ���ƫ�ߣ���a��ȷ��
b�����ȹ������о��彦ʧ�����¼�����Ľᾧˮ����ƫ������Ľᾧˮ��Ŀƫ�ߣ���b��ȷ��
c�����պ������ڿ�������ȴ���ᵼ����ˮ����ͭ���ղ���ˮ�֣�������Ľᾧˮ����ƫС���ⶨxƫС����c����
d�����ز���ʱ���������γ����Ľ��������0.1g��˵������ͭ������ȫʧȥ�ᾧˮ����Ӱ��ⶨ�������d����
�ʴ�Ϊ��ab��
�ʴ�Ϊ��������
��2������ʱ����CuSO4?xH2O�������ɫ����ɫ��ɰ�ɫʱ������ͭ����ᾧˮ��ȫʧȥ��Ӧ��ֹͣ���ȣ�
�ʴ�Ϊ����ɫ����ɫ��
��3��ֻ������ͭ�����еĽᾧˮ��ȫʧȥ������ʹ�����������ȷ��������Ҫ���к��ز�����ʹ���γ�����������С��0.1g��
�ʴ�Ϊ��ȷ������ͭ������ȫʧȥ�ᾧˮ��
��4����ɫ����ͭ������Ϊ����m4-m1��g�����ʵ���Ϊ��
| m4-m1 |
| 160 |
| m2-m4 |
| 18 |
| ||
|
| 80(m2-m4) |
| 9(m4-m1) |
�ʴ�Ϊ��
| 80(m2-m4) |
| 9(m4-m1) |
��5��a������δ����൱������ͭ�����нᾧˮ�������ⶨ���ƫ�ߣ���a��ȷ��
b�����ȹ������о��彦ʧ�����¼�����Ľᾧˮ����ƫ������Ľᾧˮ��Ŀƫ�ߣ���b��ȷ��
c�����պ������ڿ�������ȴ���ᵼ����ˮ����ͭ���ղ���ˮ�֣�������Ľᾧˮ����ƫС���ⶨxƫС����c����
d�����ز���ʱ���������γ����Ľ��������0.1g��˵������ͭ������ȫʧȥ�ᾧˮ����Ӱ��ⶨ�������d����
�ʴ�Ϊ��ab��

��ϰ��ϵ�д�
�����Ŀ