��Ŀ����
��15�֣��л��������ұ���GB2760��2011���涨���Ѿ���SO2���ʹ����Ϊ0.25g/L��ij��ȤС������9ͼ1װ�ã��г�װ���ԣ��ռ�ij���Ѿ���SO2�����Ժ������Բⶨ��

��1������A��������_______��ˮͨ��A�Ľ���Ϊ_________��
��2��B�м���300.00ml���Ѿƺ��������ᣬ����ʹSO2ȫ���ݳ�����C��H2O2��ȫ��Ӧ���仯ѧ����ʽΪ______________��
��3����ȥC�й�����H2O2��Ȼ����0.0900mol/LNaOH����Һ���еζ����ζ�ǰ������ʱ��Ӧѡ����9��ͼ2�е�_____�����ζ��յ�ʱ��Һ��pH��8.8����ѡ���ָʾ��Ϊ__________������50ml�ζ��ܽ���ʵ�飬���ζ����е�Һ���ڡ�10�����������Һ������������ţ�____���٣�10ml���ڣ�40ml���ۣ�10ml���ܣ�40ml��
��4���ζ����յ�ʱ������NaOH��Һ25.00ml�������Ѿ���SO2����Ϊ��__g/L��
��5���òⶨ�����ʵ��ֵƫ�ߣ�����ԭ����������װ������Ľ���ʩ_______��

��1������A��������_______��ˮͨ��A�Ľ���Ϊ_________��
��2��B�м���300.00ml���Ѿƺ��������ᣬ����ʹSO2ȫ���ݳ�����C��H2O2��ȫ��Ӧ���仯ѧ����ʽΪ______________��
��3����ȥC�й�����H2O2��Ȼ����0.0900mol/LNaOH����Һ���еζ����ζ�ǰ������ʱ��Ӧѡ����9��ͼ2�е�_____�����ζ��յ�ʱ��Һ��pH��8.8����ѡ���ָʾ��Ϊ__________������50ml�ζ��ܽ���ʵ�飬���ζ����е�Һ���ڡ�10�����������Һ������������ţ�____���٣�10ml���ڣ�40ml���ۣ�10ml���ܣ�40ml��
��4���ζ����յ�ʱ������NaOH��Һ25.00ml�������Ѿ���SO2����Ϊ��__g/L��
��5���òⶨ�����ʵ��ֵƫ�ߣ�����ԭ����������װ������Ľ���ʩ_______��
��1�������ܻ���������b ��2��SO2��H2O2��H2SO4 ��3���ۣ���̪���� ��4��0.24
��5��ԭ������Ļӷ����Ľ���ʩ���ò��ӷ���ǿ����������������ᣬ��������ˮ�������Ѿƽ��жԱ�ʵ�飬�۳�����ӷ���Ӱ�졣
��5��ԭ������Ļӷ����Ľ���ʩ���ò��ӷ���ǿ����������������ᣬ��������ˮ�������Ѿƽ��жԱ�ʵ�飬�۳�����ӷ���Ӱ�졣
�����������1�����������Ĺ����ص��֪������A�������ܻ�������������ʱ��ȴˮ������Ӧ�����¿ڽ����Ͽڳ�����ˮͨ��A�Ľ�����b��
��2��SO2���л�ԭ�ԣ�˫��ˮ���������ԣ�����Ϸ���������ԭ��Ӧ�������ᣬ��Ӧ�Ļ�ѧ����ʽΪSO2��H2O2��H2SO4��
��3�����������ǼӦ���ü�ʽ�ζ���ʢ�ţ���Һʱ�IJ���ѡ��ۡ��ζ��յ�ʱ��pH��8.8��˵����Һ�Լ��ԣ���ָʾ���ı�ɫ��Χ�ڼ��������£����ȵı�ɫ��Χ��3.1��4.4����̪�ı�ɫ��Χ��8.2��10.0������ѡ���ָʾ���Ƿ�̪�����ڵζ��ܵĿ̶����϶�����������������һ��û�п̶��ߣ���˸õζ�����ʵ����Һ������ǣ�50ml��10ml��40mol��
��4�����ݷ���ʽ��֪2NaOH��H2SO4��SO2����SO2��������
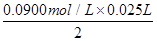 ��64g/mol��0.072g��������Ѿ���SO2�ĺ���Ϊ
��64g/mol��0.072g��������Ѿ���SO2�ĺ���Ϊ ��0.24g/L��
��0.24g/L����5�����������ǻӷ����ᣬ����Cװ�õ��������SO2����Ȼ��⣬�Ȼ���Ҳ���������Ʒ�Ӧ���Ӷ�ʹ����������������Һ��������ӣ����²ⶨ���ƫ�ߡ���˸Ľ��Ĵ�ʩΪ�ò��ӷ���ǿ�ᣬ��������������ᣬ��������ˮ�������Ѿƽ��жԱ�ʵ�飬�۳�����ӷ���Ӱ�졣

��ϰ��ϵ�д�
�����Ŀ


 (���ͪ)
(���ͪ)
 H2O + CH3CH2��O��CH2CH3 (����)
H2O + CH3CH2��O��CH2CH3 (����)



 ��3����֪C����0��1 mol Cl2�μӷ�Ӧ��������һ����������֪C�з�Ӧ�Ļ�ѧ����ʽΪ ��
��3����֪C����0��1 mol Cl2�μӷ�Ӧ��������һ����������֪C�з�Ӧ�Ļ�ѧ����ʽΪ ��


