��Ŀ����
�����з�Ӧ�У�
A.2F2+2H2O=4HF+O2
B.2Na+2H2O=2NaOH+H2��
C��CaO+H2O=Ca��OH��2
D.2H2O
2H2��+O2��
E��NO2��ˮ��Ӧ
��1��д��E��Ӧ�Ļ�ѧ����ʽ ��
��2������A��E�����Ӧ�У�ˮֻ������������ ��ˮֻ����ԭ������ ��ˮ����������������ԭ������ ��ˮ�Ȳ����������ֲ�����ԭ����������ԭ��Ӧ�� ����������ĸ��ţ�
A.2F2+2H2O=4HF+O2
B.2Na+2H2O=2NaOH+H2��
C��CaO+H2O=Ca��OH��2
D.2H2O
| ||
E��NO2��ˮ��Ӧ
��1��д��E��Ӧ�Ļ�ѧ����ʽ
��2������A��E�����Ӧ�У�ˮֻ������������
���㣺������ԭ��Ӧ
ר�⣺������ԭ��Ӧר��
��������1������������ˮ��Ӧ���������NO��
��2��ˮ�õ��ӻ��ϼ۽���ʱ����������ʧ���ӻ��ϼ�����ʱ����ԭ����ˮ��Ԫ�ػ��ϼۼ������ֽ���ʱ���������ͻ�ԭ�����Ȳ�����Ҳ������ʱ�Ȳ����������ֲ�����ԭ����
��2��ˮ�õ��ӻ��ϼ۽���ʱ����������ʧ���ӻ��ϼ�����ʱ����ԭ����ˮ��Ԫ�ػ��ϼۼ������ֽ���ʱ���������ͻ�ԭ�����Ȳ�����Ҳ������ʱ�Ȳ����������ֲ�����ԭ����
���
�⣺��1������������ˮ��Ӧ���������NO����Ӧ����ʽΪ3NO2+H2O=2HNO3+NO���ʴ�Ϊ��3NO2+H2O=2HNO3+NO��
��2��A.2F2+2H2O=4HF+O2��ˮ��OԪ�ػ��ϼ���-2�۱�Ϊ0�ۣ���ˮ����ԭ����
B.2Na+2H2O=2NaOH+H2����ˮ��HԪ�ػ��ϼ���+1�۱�Ϊ0�ۣ���ˮ����������
C��CaO+H2O=Ca��OH��2�и�Ԫ�ػ��ϼ۲��䣬����������ԭ��Ӧ��
D.2H2O
2H2��+O2����ˮ��OԪ�ػ��ϼ���-2�۱�Ϊ0�ۣ�HԪ�ػ��ϼ���+1�۱�Ϊ0�ۣ�����ˮ�������������ǻ�ԭ����
E��3NO2+H2O=2HNO3+NO��ˮ�и�Ԫ�ػ��ϼ۲��䣬NԪ�ػ��ϼ���+4�۱�Ϊ+2�ۡ�+5�ۣ�����ˮ�Ȳ����������ֲ��ǻ�ԭ����
��ˮֻ������������B��ˮֻ����ԭ������A��ˮ����������������ԭ������D��ˮ�Ȳ����������ֲ�����ԭ����������ԭ��Ӧ��E���ʴ�Ϊ��B��A��D��E��
��2��A.2F2+2H2O=4HF+O2��ˮ��OԪ�ػ��ϼ���-2�۱�Ϊ0�ۣ���ˮ����ԭ����
B.2Na+2H2O=2NaOH+H2����ˮ��HԪ�ػ��ϼ���+1�۱�Ϊ0�ۣ���ˮ����������
C��CaO+H2O=Ca��OH��2�и�Ԫ�ػ��ϼ۲��䣬����������ԭ��Ӧ��
D.2H2O
| ||
E��3NO2+H2O=2HNO3+NO��ˮ�и�Ԫ�ػ��ϼ۲��䣬NԪ�ػ��ϼ���+4�۱�Ϊ+2�ۡ�+5�ۣ�����ˮ�Ȳ����������ֲ��ǻ�ԭ����
��ˮֻ������������B��ˮֻ����ԭ������A��ˮ����������������ԭ������D��ˮ�Ȳ����������ֲ�����ԭ����������ԭ��Ӧ��E���ʴ�Ϊ��B��A��D��E��
���������⿼����������ԭ��Ӧ������Ԫ�ػ��ϼ۱仯��ϻ�������ɽ��֪������Ԫ�ػ��ϼۣ���Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ��һ������ĩ�ٷֳ�̾�ϵ�д�
��һ������ĩ�ٷֳ�̾�ϵ�д�
�����Ŀ
��ѧѧϰ�����е����Ʒ�������ѧ֪ʶͨ��Ǩ�ƹ�����֪ʶ�ķ��������������ȷ���ǣ�������
| A��п����д��Һ�е������ӷ����û���Ӧ����Ҳ������Һ�е������ӷ����û���Ӧ |
| B�������Ҵ���Ӧ���������������Ҵ�Ҳ�ܷ�Ӧ�������� |
| C��ͭ�������о���ȼ�����ɶ���ͭ��ͭҲ�����������о���ȼ�����ɶ���ͭ |
| D��ͭ��Ũ���������ȡNO2������������Ũ����Ҳ������ȡNO2 |
�������ӷ���ʽ����ȷ���ǣ�������
| A��̼�������Һ���������Լ���Һ��Ϲ��ȣ�NH4++OH-=NH3��+H2O |
| B��Fe3O4�ܽ��ڹ�����ϡ�����У�Fe3O4+8H+=Fe2++2Fe3++4H2O |
| C��������ͭ��Һ�м��������NaHS��Һ��Cu2++2HS-=CuS��+H2S�� |
| D��ʯ��ʯ�ܽ��ڴ����У�CaCO3+2H+=Ca2++H2O+CO2�� |

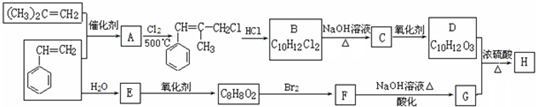
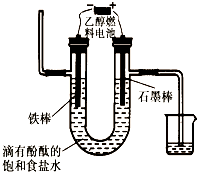 �ݱ�����һ���������ɶ�����̼�������ϳ��Ҵ��ѳ�Ϊ��ʵ��
�ݱ�����һ���������ɶ�����̼�������ϳ��Ҵ��ѳ�Ϊ��ʵ��