��Ŀ����
2������������ͭ��ҵ�������ŷ�����ܽ�Լ��Դ�����ܱ���������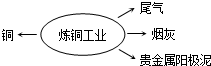
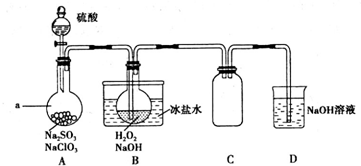
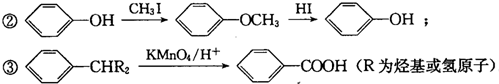
��1���ð�ˮ����β���е�SO2���ټ������ᣬ�ɻ��SO2�ͣ�NH4��2SO4���ֲ�Ʒ����ط�Ӧ�Ļ�ѧ����ʽΪSO2+2NH3•H2O=��NH4��2SO3+H2O����NH4��2SO3+H2SO4=��NH4��2SO4+SO2��+H2O��
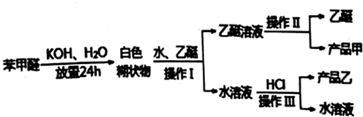
��2���̻��н���Ԫ���������κ���������̬���ڣ�����п��Ǧ�ĺ���������Ϊͭ�����ȣ��̻ҵĻ������ù������£�
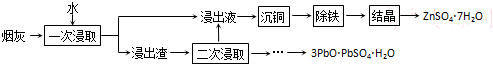
�١���ͭ���ɵõ�����ͭ���ò���Ӧ������Լ���п�ۣ������ۣ���
�ڡ�һ�ν�ȡ��ͭ�����ʽϵͣ�ԭ�����̻��в���ͭԪ����CuO���ѧʽ�������ڽ������У������ν�ȡ��Ӧѡ��H2SO4���ѧʽ������ȡ����
��3���ӹ�����������п���ȡ��������ⷨ��������ʱ����AgNO3��HNO3�Ļ��Һ�����Һ����������Ҫ�缫��ӦʽΪAg++e-=Ag������������������Ӧ����������������������Ѹ�ٱ�Ϊ����ɫ���ø���Ӧ�ĵ缫��ӦʽΪNO3-+4H++3e-=NO��+2H2O��
���� ��1��β���е�SO2�ð����գ����ð�ˮ����β���е�SO2����Ӧ����������炙���������泥�������炙���������������ᷴӦ��������李����������ˮ��
��2�����̻��н���Ԫ���������κ���������̬���ڣ����л�����п��Ǧ��ͭ����������Ƚ���ͭ���õĽ���������п�����Խ�����ͭ�û���������������ȷ�������Լ��Լ����ʵijɷ֣�
�������ʵij��ӹ����У�ע�������Լ�����������������ȷ�����ӵ��Լ���
��3�������е������������ӵĵ��ӵĻ�ԭ��Ӧ���������ӵķŵ�˳����ʵ��ԭ������д�缫��Ӧʽ��
��� �⣺β���е�SO2�ð����գ����ð�ˮ����β���е�SO2����ѧ����ʽΪ��SO2+2NH3+H2O=��NH4��2SO3����NH4��2SO3+SO2+H2O=2NH4HSO3��������Һ��NH4HSO3�ﵽһ��Ũ�Ⱥ��������ᷴӦ���ų���SO2��ѭ�����ã����ɵģ�NH4��2SO4�������ʣ���ѧ����ʽ�ǣ�2NH4HSO3+H2SO4=��NH4��2SO4+2SO2��+2H2O����NH4��2SO3+H2SO4=��NH4��2SO4+SO2��+H2O��
�ʴ�Ϊ��SO2+2NH3•H2O=��NH4��2SO3+H2O����NH4��2SO3+H2SO4=��NH4��2SO4+SO2��+H2O��
��2���̻��н���Ԫ���������κ���������̬���ڣ����л�����п��Ǧ��ͭ����������Ƚ���ͭ���õĽ���������п�����Խ�����ͭ�еĽ���ͭ�û�����������CuO�е�Cu���û������Լ������ᣬ�������������ܽ⣬���С����ν�ȡ����
�ټ���Ƚ���ͭ���õĽ���������п�����Խ�����ͭ�еĽ���ͭ�û��������ʴ�Ϊ��п�ۣ������ۣ���
���̻��в���ͭԪ����CuO���ڣ�CuO�е�Cu��������������п�û������Լ������ᣬ�������������ܽ⣬���С����ν�ȡ������Ϊ����Ҫ�������п�ľ��壬����ѡ�������ܽ�����������ʣ�
�ʴ�Ϊ��CuO��H2SO4��
��3�����AgNO3��HNO3�Ļ��Һ�ĵ����е������������ӵĵ��ӵĻ�ԭ��Ӧ���������ӵķŵ�˳�������ӵõ��ӣ��缫��ӦΪ��Ag++e-=Ag������������������Ӧ����������������������Ѹ�ٱ�Ϊ����ɫ����������NO������Ӧ������������ӵĵ��ӵĽṹ�����Ը���Ӧ�ĵ缫��ӦʽΪ��NO3-+4H++3e-=NO��+2H2O��
�ʴ�Ϊ��Ag++e-=Ag��NO3-+4H++3e-=NO��+2H2O��
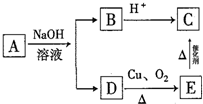
���� �����漰���ʵķ�����ᴿ�����صĹ���ԭ���Լ��缫��Ӧʽ����д֪ʶ�������ۺ�֪ʶ�Ŀ��飬�Ѷ��еȣ�

| A�� | ���Ʊ�С�մ�ķ���ʽΪ��NaCl+NH4HCO3��NaHCO3��+NH4Cl | |
| B�� | ĸҺ��ͨ��İ�����HCO3-��Ӧ��NH3+HCO3-��CO32-+NH4+ | |
| C�� | ��ʳ����Ϊ������Һ��Cl-��Ũ�� | |
| D�� | �����֪�¶Ƚϵ�ʱ���Ȼ�淋��ܽ�ȱ��Ȼ��ƵĴ� |
| A�� | ������Ũ��������������棬����Ϊ���������Ũ�����жۻ� | |
| B�� | ����������Һ����������Ƥ���IJ����Լ�ƿ�� | |
| C�� | �������еμ�Ũ���ᣬ���DZ������ΪŨ���������ˮ�� | |
| D�� | Ũ���ᱣ������ɫ�����Լ�ƿ�� |
| A�� | 1�� | B�� | 2�� | C�� | 3�� | D�� | 4�� |
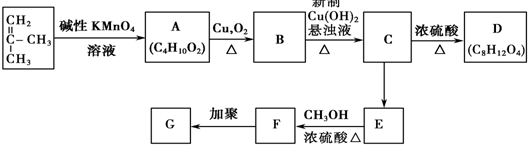
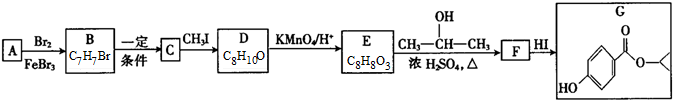
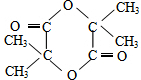 ��G�Ľṹ��ʽ��
��G�Ľṹ��ʽ��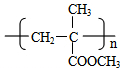 ��
��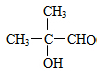 +3Cu��OH��2$\stackrel{��}{��}$
+3Cu��OH��2$\stackrel{��}{��}$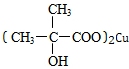 +Cu2O+4H2O��
+Cu2O+4H2O��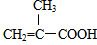 +CH3OH$��_{��}^{ŨH_{2}SO_{4}}$
+CH3OH$��_{��}^{ŨH_{2}SO_{4}}$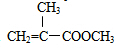 +H2O��
+H2O��
 ��
��
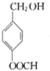
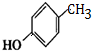 G�Ĺ���������Ϊ�ǻ�������
G�Ĺ���������Ϊ�ǻ�������
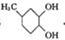

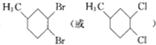 ��
��