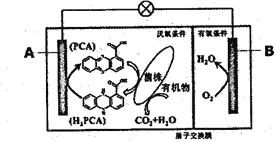
ΧβΡΩΡΎ»ί
ΓΨΧβΡΩΓΩΡ≥Μ·―ß–Υ»Λ–ΓΉιΕ‘2-δε±ϊΆι”κNaOH»ή“ΚΖΔ…ζΒΡΖ¥”ΠΫχ––ΧΫΨΩΘ§ Β―ιΉΑ÷Ο»γœ¬Θ®Φ–≥÷ΚΆΦ”»»ΉΑ÷Ο“―¬‘»ΞΘ©ΘΚ

Β―ι≤Ϋ÷ηΘΚ
ΔΓΘ°ΫΪ‘≤ΒΉ…’ΤΩ”ΟΥ°‘ΓΦ”»»Θ§άδΡΐΙή÷–Ά®»κάδΡΐΥ°ΘΜ
ΔΔΘ°ΫΪΖ÷“Κ¬©ΕΖ÷–ΒΡ2-δε±ϊΆι”κNaOH»ή“ΚΒΡΜλΚœ“ΚΒΈ»κ‘≤ΒΉ…’ΤΩΡΎΓΘ
Θ®1Θ©Υ°άδΡΐΙήaΩΎ «__________Θ®ΧνΓΑΫχΥ°ΩΎΓ±ΜρΓΑ≥ωΥ°ΩΎΓ±Θ©ΓΘ
Θ®2Θ©»ΓΨΏ÷ß ‘Ιή÷– ’Φ·ΒΫΒΡ“ΚΧεΘ§Ψ≠ΚλΆβΙβΤΉΦλ≤βΘ§Τδ÷–“Μ÷÷Έο÷ ÷–¥φ‘ΎC-OΦϋΚΆO-HΦϋΓΘ‘ρΗΟΈο÷ ΒΡΫαΙΙΦρ Ϋ «_________________ΓΘ
Θ®3Θ© Β―ι÷–Υα–‘KMnO4»ή“ΚΆ …ΪΘ§…ζ≥…ΗΟ ΙΥα–‘KMnO4»ή“ΚΆ …ΪΒΡΈο÷ ΒΡΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ Ϋ «____________________________________________________________ΓΘ
Θ®4Θ©Ά®ΙΐΗΟ Β―ιΒΟΒΫΒΡΫα¬έ «___________________________________________________ΓΘ
ΓΨ¥πΑΗΓΩ ≥ωΥ°ΩΎ (CH3)2CHOH ![]() 2-δε±ϊΆι”κ«β―θΜ·ΡΤ»ή“ΚΦ»ΡήΖΔ…ζ»Γ¥ζΖ¥”Π”÷ΡήΖΔ…ζœϊ»ΞΖ¥”ΠΘ§ΝΫΗωΖ¥”Π‘ΎΆ§“ΜΧθΦΰœ¬Ά§ ±ΖΔ…ζ
2-δε±ϊΆι”κ«β―θΜ·ΡΤ»ή“ΚΦ»ΡήΖΔ…ζ»Γ¥ζΖ¥”Π”÷ΡήΖΔ…ζœϊ»ΞΖ¥”ΠΘ§ΝΫΗωΖ¥”Π‘ΎΆ§“ΜΧθΦΰœ¬Ά§ ±ΖΔ…ζ
ΓΨΫβΈωΓΩΘ®1Θ©ΈΣΝΥ≥δΖ÷ά以’τΤχΘ§±Ί–κ ΙάδΡΐΙήΡΎ≥δ¬ζάδ»¥Υ°Θ§Ι –η“ΣΒΆΫχΗΏ≥ωΘ§άδΡΐΙήaΩΎ «≥ωΥ°ΩΎΘΜ(2) 2-δε±ϊΆι”κNaOH»ή“ΚΖ¥”ΠΒΡ…ζ≥…ΈοΫαΙΙ÷–¥φ‘ΎC-OΦϋΚΆO-HΦϋΘ§‘ρΥΒΟς2-δε±ϊΆι‘Ύ¥ΥΧθΦΰΖΔ…ζΝΥΥ°ΫβΖ¥”Π…ζ≥…ΝΥ(CH3)2CHOHΚΆδεΜ·ΡΤΘΜΘ®3Θ©Μ”ΖΔ≥ωά¥ΒΡ““¥ΦΨ≠Υ°œ¥Κσ»ήΫβ‘ΎΥ°÷–Θ§2-δε±ϊΆιΖ–ΒψΫœΗΏΨ≠ά以Κσ≤ΜΜαΫχ»κΥ°÷–Θ§“Σ ΙΥα–‘KMnO4»ή“ΚΆ …ΪΘ§Ω…Ρή «‘Ύ«ΑΟφΖ¥”Π÷–ΜΙ…ζ≥…ΝΥΡ―»ή”ΎΥ°«“Ζ–ΒψΒΆΒΡœ©ΧΰΘ§Υυ“‘Ζ¥”ΠΖΫ≥Χ ΫΈΣΘΚ![]() ΘΜ
ΘΜ
Θ®4Θ©Ά®Ιΐ…œ ω Β―ιΥΒΟς2-δε±ϊΆι”κ«β―θΜ·ΡΤ»ή“ΚΦ»ΡήΖΔ…ζ»Γ¥ζΖ¥”Π”÷ΡήΖΔ…ζœϊ»ΞΖ¥”ΠΘ§ΝΫΗωΖ¥”Π‘ΎΆ§“ΜΧθΦΰœ¬Ά§ ±ΖΔ…ζΓΘ

 ΟΩΩΈ±ΊΝΖœΒΝ–¥πΑΗ
ΟΩΩΈ±ΊΝΖœΒΝ–¥πΑΗ «…―ß«…ΝΖœΒΝ–¥πΑΗ
«…―ß«…ΝΖœΒΝ–¥πΑΗΓΨΧβΡΩΓΩœ¬±μ «ΦΗ÷÷»θΒγΫβ÷ ΒΡΒγάκΤΫΚβ≥Θ ΐ (25Γφ)ΓΘ
ΒγΫβ÷ | ΒγάκΖΫ≥Χ Ϋ | ΤΫΚβ≥Θ ΐK |
CH3COOH | CH3COOH | 1.76 ΓΝ 10 -5 |
H2CO3 | H2CO3 HCO3ΓΣ | K1=4.31 ΓΝ 10 -7 K2=5.61 ΓΝ 10 -11 |
C6H5OH | C6H5OH | 1.1 ΓΝ 10 -10 |
H3PO4 | H3PO4 H2PO4ΓΣ HPO42ΓΣ | K1=7.52 ΓΝ 10 -3 K2=6.23ΓΝ 10 -8 K3=2.20ΓΝ 10 -13 |
NH3ΓΛH2O | NH3ΓΛH2O | 1.76ΓΝ 10 -5 |
ΜΊ¥πœ¬Ν–Έ ΧβΘ®C6H5OHΈΣ±ΫΖ”Θ©ΘΚ
Θ®1Θ©”……œ±μΖ÷ΈωΘ§»τ ΔΌ CH3COOH ΔΎ HCO3ΓΣ Δέ C6H5OH Δή H2PO4ΓΣ ΨυΩ…Ω¥ΉςΥαΘ§‘ρΥϋΟ«Υα–‘”…«ΩΒΫ»θΒΡΥ≥–ρΈΣ___________Θ®Χν±ύΚ≈Θ©ΘΜ
Θ®2Θ©–¥≥ωC6H5OH”κNa3PO4Ζ¥”ΠΒΡάκΉ”ΖΫ≥Χ ΫΘΚ__________________________ΘΜ
Θ®3Θ©25Γφ ±Θ§ΫΪΒ»ΧεΜΐΒ»≈®Ε»ΒΡ¥ΉΥαΚΆΑ±Υ°ΜλΚœΘ§ΜλΚœ“Κ÷–ΘΚc(CH3COOΓΣ)______c(NH4+)ΘΜΘ®ΧνΓΑΘΨΓ±ΓΔΓΑΘΫΓ±ΜρΓΑΘΦΓ±Θ©
Θ®4Θ©25Γφ ±Θ§œρ10 mL 0.01 mol/L±ΫΖ”»ή“Κ÷–ΒΈΦ”V mL 0.01 mol/LΑ±Υ°Θ§ΜλΚœ»ή“Κ÷–ΝΘΉ”≈®Ε»ΙΊœΒ’ΐ»ΖΒΡ «______ΘΜ
A.»τΜλΚœ“ΚpHΘΨ7Θ§‘ρVΓί 10
B.»τΜλΚœ“ΚpHΘΦ7Θ§‘ρc((NH4+) ΘΨc (C6H5OΓΣ) ΘΨc (H+)ΘΨc (OHΓΣ)
C.V=10 ±Θ§ΜλΚœ“Κ÷–Υ°ΒΡΒγάκ≥ΧΕ»–Γ”Ύ10 mL 0.01mol/L±ΫΖ”»ή“Κ÷–Υ°ΒΡΒγάκ≥ΧΕ»
DΘ°V=5 ±Θ§2c(NH3ΓΛH2O)+ 2 c (NH4+)= c (C6H5OΓΣ)+ c (C6H5OH)
Θ®5Θ©Υ°ΫβΖ¥”Π «Βδ–ΆΒΡΩ…ΡφΖ¥”ΠΘ°Υ°ΫβΖ¥”ΠΒΡΜ·―ßΤΫΚβ≥Θ ΐ≥ΤΈΣΥ°Ϋβ≥Θ ΐ(”ΟKh±μ Ψ)Θ§άύ±»Μ·―ßΤΫΚβ≥Θ ΐΒΡΕ®“εΘ§«κ–¥≥ωNa2CO3ΒΎ“Μ≤ΫΥ°ΫβΖ¥”ΠΒΡΥ°Ϋβ≥Θ ΐΒΡ±μ¥ο Ϋ_______________ΓΘ