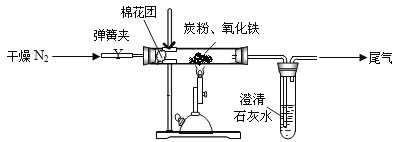
��Ŀ����
����Ŀ��X��Y��Z��U��W��ԭ���������������ǰ������Ԫ�ء�����Y��ԭ�Ӻ�����7���˶�״̬��ͬ�ĵ��ӣ�X��Z��δ�ɶԵ�������Ϊ2��U�ǵ�������Ԫ���γɵļ������а뾶��С��Ԫ�أ�W���ڲ����ȫ�����������ֻ��1�����ӡ���ش��������⣺
(1)X��Y��Z�ĵ�һ�����ܴӴ�С��˳����_______ (��Ԫ�ط��ű�ʾ����ͬ)��
(2)д��W�ļ۵����Ų�ʽ________ ��Wͬ���ڵ�Ԫ���У���Wԭ��������������ȵ�Ԫ�ػ���_______��
(3)���ݵȵ�����ԭ������֪������XZ�Ľṹʽ��______��YZ2-��VSEPRģ����________��
(4)X��Y��Z�ļ��⻯��ļ��ǴӴ�С��˳����________(�û�ѧʽ��ʾ)��ԭ����____________________��
(5)��Ԫ��Y��UԪ����ɵĻ�����A�������ṹ��ͼ��ʾ�������ʾYԭ�ӣ������ʾUԭ�ӣ�����д��������A�Ļ�ѧʽ_______��������Ӳ�ȴ��Ʋ������Ϊ______���塣����YԪ�ص��ӻ���ʽ��____________��

(6)U�ľ�����������ϵ���侧���߳�Ϊ405Pm���ܶ���2.70g��cm-3��ͨ������ȷ���侧��������________________���Ѽ������ѻ������������ѻ��������������ܶѻ�������֪��4053=6.64��107)��
���𰸡� N>O>C 3d104s1 K��Cr C![]() O ƽ�������� CH4>NH3>H2O 3���⻯�������ԭ�Ӽ۲���Ӷ��پ�Ϊ4��VSEPRģ�;�Ϊ��������ṹ��������ԭ�ӵŵ��Ӷ����������ӣ����¼��DZ�С AlN ԭ�� sp3 �����������ܶѻ�
O ƽ�������� CH4>NH3>H2O 3���⻯�������ԭ�Ӽ۲���Ӷ��پ�Ϊ4��VSEPRģ�;�Ϊ��������ṹ��������ԭ�ӵŵ��Ӷ����������ӣ����¼��DZ�С AlN ԭ�� sp3 �����������ܶѻ�
��������X��Y��Z��U��W��ԭ���������������ǰ������Ԫ�ء�����Y��ԭ�Ӻ�����7���˶�״̬��ͬ�ĵ��ӣ���YΪ��Ԫ�أ�U�ǵ�������Ԫ���γɵļ������а뾶��С��Ԫ�أ���UΪ��Ԫ�أ�X��Z��δ�ɶԵ�������Ϊ2��X��ԭ������С��N����Z��ԭ����������N������֮�䣻 X��Z�ĺ�������Ų��ֱ�Ϊ1s22s22p2��1s22s22p4����XΪ̼Ԫ�ء�ZΪ��Ԫ�أ�W���ڲ����ȫ�����������ֻ��1�����ӣ�ԭ��������������ֻ�ܴ��ڵ������ڣ����������Ϊ2+8+18+1=29����WΪͭԪ�ء���1��ͬ����Ԫ����ԭ����������һ�����ܳ��������ƣ���NԪ��2p�ܼ�Ϊ�����ȶ�״̬�������ϵͣ���һ�����ܸ���ͬ��������Ԫ�أ��ʵ�һ������N>O>C����2��WΪͭԪ�أ��۵����Ų�ʽΪ3d104s1��Wͬ���ڵ�Ԫ���У���Wԭ��������������ȵ�Ԫ�ػ���3d64s1��ΪKԪ�ء�3d54s1ΪCrԪ�أ���3�����ݵȵ�����ԭ������֪������CO��N2��Ϊ�ȵ����壬CO�Ľṹʽ��C![]() O��YZ2��ΪNO2��������ԭ�Ӽ۵��Ӷ���=2+
O��YZ2��ΪNO2��������ԭ�Ӽ۵��Ӷ���=2+![]() ����5+1-2��2��=3��VSEPRģ����ƽ�������Σ���4��X��Y��Z������⻯��ֱ�ΪCH4��NH3��H2O�������⻯�������ԭ�Ӽ۲���Ӷ�����Ϊ4��VSEPRģ�;�Ϊ���������Σ�������ԭ�ӵŵ��Ӷ������������¼��DZ�С���ʼ��ǣ�CH4>NH3>H2O����5��������Nԭ����ĿΪ4����ԭ����ĿΪ8��
����5+1-2��2��=3��VSEPRģ����ƽ�������Σ���4��X��Y��Z������⻯��ֱ�ΪCH4��NH3��H2O�������⻯�������ԭ�Ӽ۲���Ӷ�����Ϊ4��VSEPRģ�;�Ϊ���������Σ�������ԭ�ӵŵ��Ӷ������������¼��DZ�С���ʼ��ǣ�CH4>NH3>H2O����5��������Nԭ����ĿΪ4����ԭ����ĿΪ8��![]() +6��
+6��![]() =4���ʸû����ﻯѧʽΪAlN��������Ӳ�ȴ�����ԭ�Ӿ��壬Yԭ���γ�4�����ۼ�����Yԭ���ӻ���ʽΪsp3����6���辧������ԭ����ĿΪN(Al)����N(Al)=
=4���ʸû����ﻯѧʽΪAlN��������Ӳ�ȴ�����ԭ�Ӿ��壬Yԭ���γ�4�����ۼ�����Yԭ���ӻ���ʽΪsp3����6���辧������ԭ����ĿΪN(Al)����N(Al)=![]() g=(405��10-10)3��2.70g/cm3�����N(Al)=4.00�������侧��������Ϊ���������ܶѻ���
g=(405��10-10)3��2.70g/cm3�����N(Al)=4.00�������侧��������Ϊ���������ܶѻ���

 ����ѵ�����⿼ϵ�д�
����ѵ�����⿼ϵ�д� �������ϵ�д�
�������ϵ�д�����Ŀ������ת��,��Ҫ����������������ɵ���( )
A. H2SO4 �� SO2 B. Fe ��FeCl2 C. MnO4-�� Mn2+ D. CO2��CO32-
����Ŀ����ҵȼ��ú��ʯ�͵Ȼ�ʯȼ���ͷų�������������(NOx)��CO2��SO2�����壬������Ⱦ�������Է���������������̼����������ʵ����ɫ�������������á�
��.������
��֪��H2��ȼ����Ϊ285.8kJ��mol-1
N2(g)+2O2(g)��2NO2(g)��H��+133kJ��mol-1
H2O(g)��H2O(l) ��H��-44kJ��mol-1
���������£�H2��ԭNO2����ˮ���������������ʵ��Ȼ�ѧ����ʽΪ��____________��
��.��̼��
(1)��2L�ܱ������м���2molCO2��6molH2�����ʵ��Ĵ��������£�������Ӧ��
CO2(g)+3H2(g)![]() CH3OH(l)+H2O(l)
CH3OH(l)+H2O(l)
�ٸ÷�Ӧ�Է����е�������_____________(����¡��������¡��������¶ȡ�)
������������˵���˷�Ӧ�ﵽƽ��״̬����____________��(����ĸ)
a.��������ƽ��ʽ�����ֲ��� b.CO2��H2������������ֲ���
c.CO2��H2��ת������� d.���������ܶȱ��ֲ���
e.1molCO2���ɵ�ͬʱ��3mol H��H������
��CO2��Ũ����ʱ�䣨0��t2���仯����ͼ��ʾ����t2ʱ�������ݻ���Сһ����t3ʱ�ﵽƽ�⣬t4ʱ�����¶ȣ�t5ʱ�ﵽƽ�⣬�뻭��t2��t6 CO2Ũ����ʱ��ı仯��_____________
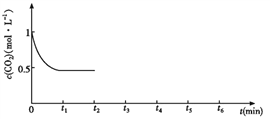
�Ƹı��¶ȣ�ʹ��ӦCO2(g)+3H2(g)![]() CH3OH(g)+H2O(g) ��H��0�е��������ʶ�Ϊ��̬����ʼ�¶ȡ������ͬ��T1�桢2L�ܱ�����������Ӧ�����в������ݼ��±���
CH3OH(g)+H2O(g) ��H��0�е��������ʶ�Ϊ��̬����ʼ�¶ȡ������ͬ��T1�桢2L�ܱ�����������Ӧ�����в������ݼ��±���
��Ӧʱ�� | CO2(mol) | H2(mol) | CH3OH(mol) | H2O(mol) | |
��Ӧ���º��� | 0min | 2 | 6 | 0 | 0 |
10min | 4.5 | ||||
20min | 1 | ||||
30min | 1 | ||||
��Ӧ���Ⱥ��� | 0min | 0 | 0 | 2 | 2 |
�ٴﵽƽ��ʱ����Ӧ��Աȣ�ƽ�ⳣ��K(I)______K(II)�������������������ͬ����ƽ��ʱCH3OH��Ũ��c(I)____ c(II)��
�ڶԷ�Ӧ��ǰ10min�ڵ�ƽ����Ӧ����v(CH3OH)��_______���������������������£���30minʱֻ�ı��¶�T2�棬��ʱH2�����ʵ���Ϊ3.2mol����T1___T2(�>������<����=��)����30minʱֻ���������ٳ���1molCO2(g)��1molH2O(g)����ƽ��_____�ƶ�(�����������)��
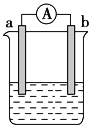
�������˹�������ÿɽ�CO2ת��Ϊ���ᣬ��Ӧԭ��Ϊ2CO2+2H2O=2HCOOH+O2,
װ����ͼ��ʾ��
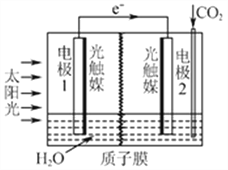
�ٵ缫2�ĵ缫��Ӧʽ��____________��
���ڱ�״���£����缫2����11.2L CO2��Ӧ�� �����ϵ缫1��Һ������_____(����ӡ����١�______g��