��Ŀ����
���������е�+4�����������ԣ����л�ԭ�ԡ������Լ�����ˮ��Na2S��Һ�� Na2SO3��Һ��ϡ���ᡢNaOH��Һ����ˮ��
(1)Ҫ֤��Na2SO3���л�ԭ�ԣ�Ӧѡ�õ��Լ��Уߣߣߣߣߣ������������ǣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߡ�
��Ӧ�����ӷ���ʽΪ���ߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߡ�
(2)Ҫ֤��Na2SO3���������ԣ�Ӧѡ�õ��Լ��Уߣߣߣߣߣ������������ǣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߡ�
��Ӧ�����ӷ���ʽΪ���ߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߡ�
(1)��ˮ��Na2SO3��Һ����������ˮ��ɫ����ȥ SO32-+Br2+H2O=SO42-+2Br-+2H+
(2)Na2S��Һ��Na2SO3��Һ��ϡ���� �е���ɫ�ij������� SO32-+2S2-+6H+=3S��+3H2O
����

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�ijͬѧ������ͼװ����NO���������ʵ��̽����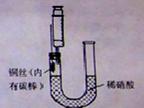
��1��д���Ʊ�NO�ķ�Ӧ����ʽ
��2���ռ��������������Ϻ���ɫ����˵��NO����_____�ԡ�
��3���Ʊ�NO�IJ������£�
1��ͼ��ʾ���Ӻ�װ�ã�
2ȡ��ע������ͨ��U�ͳ��ܼ���ϡ�������ӽ�U�Ͷ̹ܵ�ͭ˿
3����ͷ�ϰ�װ��ע���������U�Ͷ̹��е����п�����Ȼ��γ�ע���������ž�ע�����еĿ�����
4��������ͭ˿����������ų�������ע�����������NO����ע������ͷ������Ƥ�����Է�ڡ�
��������������©��һ�������� ���ڲ���3�п��Թ۲쵽������Ϊ ��
��4����ͭ˿�ϰ���ʯī�������� ��
��5������ϡ���ỻ��Ũ���ᣬ�����ȡNO2���塣������ע�����ռ�NO2���岢��ɡ�������ضԻ�ѧƽ���ƶ�Ӱ�족ʵ��̽����������������������ֻ���һ��Ӱ�����ص�̽��������ʵ����Ʒ��ѡ����֪
2NO2(g)  N2O4(g) ��H ��0��N2O4Ϊ��ɫ���塣
N2O4(g) ��H ��0��N2O4Ϊ��ɫ���塣
| ʵ�鲽�衡�� | ʵ������ | ʵ����ۡ��� |
| �ٷֱ�����֧ע�����ռ�NO2������ע������ͷ������Ƥ�����Է�ڡ����� ��_____________________���� _______________________���� ________________________���� _______________________���� ________________________���� | _______________________���� ________________________���� _______________________���� ________________________���� _______________________���� _______________________���� ________________________���� _______________________���� | _______________________���� ________________________���� _______________________���� ________________________���� _______________________���� _______________________���� ________________________���� _______________________���� |

 ���� ������+��Cl-+��H2O
���� ������+��Cl-+��H2O
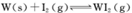 ��Ϊģ��������Ӧ��ȷ��ȡ0. 508g�⡢0.736g����������50. 0mL���ܱ������У�����ʹ�䷴Ӧ����ͼ�� WI2(g)�����ʵ�����ʱ��仯��ϵͼ����������I��0��t2ʱ��Σ��ķ�Ӧ�¶�ΪT1������II(��t2��ʼ)�ķ�Ӧ�¶�ΪT2����T2>T1����
��Ϊģ��������Ӧ��ȷ��ȡ0. 508g�⡢0.736g����������50. 0mL���ܱ������У�����ʹ�䷴Ӧ����ͼ�� WI2(g)�����ʵ�����ʱ��仯��ϵͼ����������I��0��t2ʱ��Σ��ķ�Ӧ�¶�ΪT1������II(��t2��ʼ)�ķ�Ӧ�¶�ΪT2����T2>T1����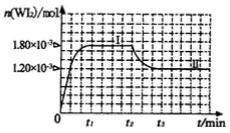

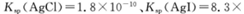
 ]��
]��
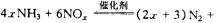
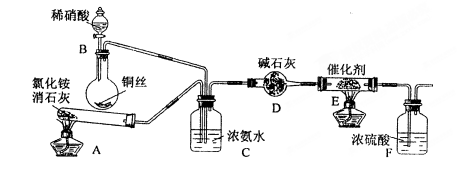
 ��ij��ѧ��ȤС��ģ��ô������̵�ʵ��װ�����£��г�װ������ȥ����
��ij��ѧ��ȤС��ģ��ô������̵�ʵ��װ�����£��г�װ������ȥ����