��Ŀ����
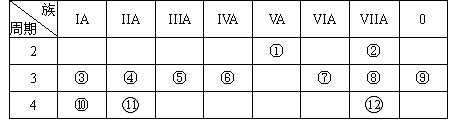
���ֶ�����Ԫ��A��B��C��D��E��ԭ��������������A��Cͬ�壬B��D ͬ�壬C���Ӻ�B���Ӿ�����ͬ�ĵ��Ӳ�ṹ��A��B��D��E�����γɹ����ͻ����A��B�γɵ�������ˮ�гʼ��ԣ�C��E�γɵĻ�������ˮ�г����ԡ��ش��������⣺
��1������Ԫ���У�ԭ�Ӱ뾶������ ���ǽ�������ǿ���� ����Ԫ�ط��ţ���
��2����A��B��D��E���γɵĹ����ͻ������У����ȶ��������� ���û�ѧʽ��ʾ����
��3��A��E�γɵĻ�������A��B�γɵĻ����ﷴӦ������Ļ�ѧʽΪ �����д��ڵĻ�ѧ������Ϊ ��
��4��D����������ˮ����Ļ�ѧʽΪ ��
��5������E��ԭ�ӽṹʾ��ͼΪ ��D�ڲ������E��ȼ�գ����ɵ���Ҫ����Ļ�ѧʽΪ ��
��6������E��ˮ��Ӧ�����ӷ���ʽΪ ��
��1��Na �� Cl
��2��PH3
��3��NH4Cl
��4��H3PO4
��5�� ��PCl3
��PCl3
��6��Cl2+H2O=HClO+H++Cl-
�������������A��B�γɵ�������ˮ�гʼ��ԣ�˵��A��H��B��NԪ�أ�A��B��C��D��E��ԭ������������������C��Na��D��PԪ�أ�C��E�γɵĻ�������ˮ�г����ԣ�E��ClԪ�ء�
��1������Ԫ���У�ԭ�Ӱ뾶������Na���ǽ�������ǿ����Cl��
��2��B��D��E�зǽ�������������P��������A�γɵ���̬�⻯�����ȶ�������PH3
��3��A��E�γɵĻ��������Ȼ��⣬A��B�γɵĻ������ǰ��������߷�Ӧ�����Ȼ�泥���ѧʽΪNH4Cl
(4) D����������ˮ���������ᣬ��ѧʽΪH3PO4
��5���ȵ�ԭ�ӽṹʾ��ͼΪ ��P�ڲ����������ȼ�գ��������Ȼ��ף���ѧʽΪPCl3
��P�ڲ����������ȼ�գ��������Ȼ��ף���ѧʽΪPCl3
��6��������ˮ��Ӧ�����ӷ���ʽΪCl2+H2O=HClO+H++Cl-
���㣺����Ԫ���ƶϣ�ԭ�Ӱ뾶���ǽ����ԡ���̬�⻯����ȶ��ԡ���ѧʽ��ԭ�ӽṹʾ��ͼ�����ӷ���ʽ���ж�

��12�֣�����ѧ�������ʽṹ�����ʡ�
����Ԫ�ذ���N��P��As��Sb��Bi����Ԫ�ء�
��1�����й��ڵ���Ԫ�ص�˵����ȷ���� ��
| A��N2�����������ơ��䷢��ԭ���ǵ��Ӵ������ϵ͵Ĺ��ԾǨ�������ϸߵĹ�����Թ����ʽ�ͷ����� |
| B��P��Na��S����Ԫ�صĵ�һ�������ɴ�С��˳���ǣ�P>S>Na |
| C����̬Asԭ���У�����ռ�ݵ�����ܼ�Ϊ4d |
| D��Biԭ�����������5��������ͬ�ĵ��� |
��3��PCl3�����У�Cl��P��Cl���ļ���____ 109o28�����>����<����=������
��4�����ף�P4������һ�ֵ��ʣ������ڷ��Ӿ��壬�侧���ṹ��ͼ����֪�þ�����ܶ�Ϊ
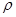 g��cm��3�������ı߳�Ϊacm�����ӵ�����Ϊ__ mol-l���ú��ѡ�a��ʽ�ӱ�ʾ����
g��cm��3�������ı߳�Ϊacm�����ӵ�����Ϊ__ mol-l���ú��ѡ�a��ʽ�ӱ�ʾ����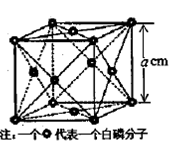
�±����г����ֶ�����Ԫ��A��B��C��D��E����Ϣ�����ƶϺ�ش�
| Ԫ�� | �� �� �� Ϣ |
| A | Ԫ����Ҫ���ϼ�Ϊ-2��ԭ�Ӱ뾶Ϊ0.074 nm |
| B | ��������������������������֮��Ϊ4 |
| C | ԭ�Ӱ뾶Ϊ0.102 nm���䵥��Ϊ��ɫ���壬����A�ĵ�����ȼ�� |
| D | ����������ˮ�����ܰ�1�U1�������������ȵ����������� |
| E | ԭ�Ӱ뾶Ϊ0.075 nm������������ˮ����������⻯���γ�һ����X |
��1��д��CԪ�������ڱ��е�λ���� ��д��DԪ������������ˮ�������ʽ������ ��
��2��д��B������ˮ��Ӧ�����ӷ���ʽ ��
��3��Ԫ��A��D�γɵ�ij�ֻ��������Ϊ�����������������Դ��д���õ�������Ӧ����Ҫ��ѧ����ʽ ��
��4��X��ˮ��Һ������ ������ᡱ��������С����ԣ������ӷ���ʽ������ԭ�������� ��
��5����֪EԪ�ص�ij���⻯��Y��A2��Ħ��������ͬ��Y�������ɵ�ȼ�ϵ���У��������Һ��30����KOH��Һ���õ�طŵ�ʱ�����ĵ缫��ӦʽΪ ��
��6����ʹ��Y������ȼ�ϵ�ؾ���ͭ�����õ���ͭ80 gʱ��ȼ�ϵ����ת�Ƶĵ�����Ϊ NA��
I����Ԫ�ط��Żش�ԭ������11-18��Ԫ�ص��й����⣺(���Ӧ���ʵĻ�ѧʽ)
��1������������ˮ���������ǿ���� ;�����ʽΪ��
��2������������ˮ��������Ե��� ;
��3�����γ���̬�⻯�������ȶ����� �������ʽΪ ��
II����ѧ��һ����ʵ��Ϊ��������Ȼ��ѧ����������ʵ��֪ʶ�ش��������⡣
����˵��������� ��
| A����Cl2ͨ����ɫʯ����Һ����Һ�ȱ�����ɫ |
| B����Һʱ����Һ©�����²�Һ����¿ڷų����ϲ�Һ����Ͽڵ��� |
| C������1L0��5mol��L-1��NaCl��Һ������������ӳ���29��25gNaCl���� |
| D�������£�������������������Ũ���� |
F��ʵ���ұ�������������Һ�������Լ�ƿ�ڼ������ۻ���������ֹ��������������
G����ȥKCl��Һ������MgCl2����������NaOH��Һ������