��Ŀ����
���Ӽ�����������Ŧ��ʽ���Ϊ��п��أ���缫�ֱ�ΪAg2O��Zn���������ҺΪ KOH��Һ���缫��ӦΪ Zn+2OH-2e-=Zn(OH)2�� Ag2O+H2O+2e-=2Ag+2OH-���ܷ�ӦʽΪ Ag2O+H2O +Zn = Zn(OH)2+2Ag��������������ȷ����
A��������п�����·����������
B���õ�ؿ�ʵ�ֻ�ѧ�ܺ͵��ܵ��ת��
C��п�Ǹ�����������������
D������ʱ����������Һ pH ��С�������� pH ����
��ϰ��ϵ�д�
�����Ŀ




 װ�ã���Ӧǰ���缫������ȣ�һ��ʱ������缫�������12 g��������ͨ��________mol���ӡ�
װ�ã���Ӧǰ���缫������ȣ�һ��ʱ������缫�������12 g��������ͨ��________mol���ӡ�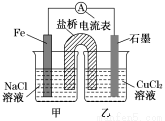

 FeO42?+3H2��������ԭ������ͼ1��ʾ��װ��ͨ������缫���������Ϻ�ɫ��FeO42?�����缫�����ݲ�����������������ҺŨ�ȹ��ߣ����缫����������ɫ���ʡ���֪��Na2FeO4ֻ��ǿ�����������ȶ����ױ�H2��ԭ��
FeO42?+3H2��������ԭ������ͼ1��ʾ��װ��ͨ������缫���������Ϻ�ɫ��FeO42?�����缫�����ݲ�����������������ҺŨ�ȹ��ߣ����缫����������ɫ���ʡ���֪��Na2FeO4ֻ��ǿ�����������ȶ����ױ�H2��ԭ��

 ����ҵ�ϳ�·�����£�
����ҵ�ϳ�·�����£�

 �����ɳ�����������NaOH��Һ����� mL��
�����ɳ�����������NaOH��Һ����� mL��