��Ŀ����
����Ŀ�����������һ����;�㷺�ij�������������Ҫ���ڻ�������������ҩ��ҵ�ȡ�
I.ij����С�������������Ʊ�������صķ������£�
i.����ص��Ʊ���
(1)���������̡�����غ��������ع������______(����ĸ)������������ء�
A���ձ�B��������C��������D��������
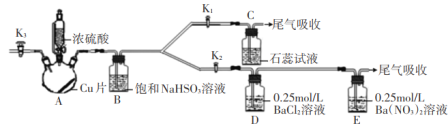
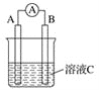
ii.������ص��Ʊ�(װ����ͼ��ʾ�����ȼ��г�װ��ʡ��)��
��i���Ƶõ�����ؼ���������ˮ�ܽ⣬��ī��ɫ��Һ������������ƿ�С���װ���еķ�Һ©����������ϡ���ᣬ���ȣ�ʹ���ɵ����������װ���в�ˮԡ���ȡ������װ���е���Һ������������������·�Ӧ���ɸ�����غͶ������̡�
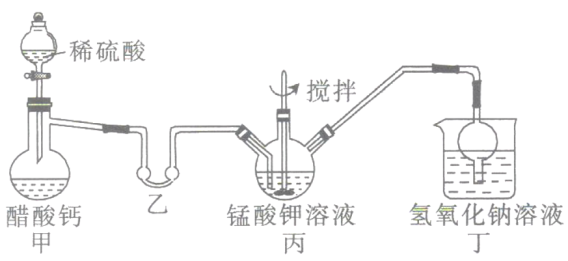
(2)�����װ�������Եķ��������Ӻ�װ�ã�_________________��
(3)��װ�õ�������___________����װ����NaOH��Һ��������____________________��
(4)��������ƿ�����ò�����й���,����Һ�����������У�_______��______�����ˣ��õ���״�ĸ�����ؾ��塣������ؾ���ֻ�ܵ��º�ɣ���ԭ��Ϊ_____________________��
II.����������ԭ�ζ����ⶨ������صĴ��ȣ�������Ӧ�Ļ�ѧ����ʽΪ2KMnO4+5H2C2O4+3H2SO4=K2SO4+2MnSO4+10CO2��+8H2O��
(5)��ȡһ������ĸ��������Һ��ʹ�õζ���_______(����ĸ)��

(6)����ʵ������д������в�������ʹ����KMnO4Ũ��ƫ�ߵ���____(����ĸ)��
A��ƿ��ˮϴ֮��ĩ�ô���Һ��ϴ
B.ʢ������Һ�ĵζ��ܵζ�ǰ���첿�������ݣ��ζ��յ�ʱ������
C.�ζ�ʱ��������Һ����ƿ��
D.ʢװKMnO4��Һ�ĵζ�����ˮϴ��δ��KMnO4��Һ��ϴ
���𰸡�D �رշ�Һ©�����������ձ��м���ˮ���þƾ�����������ƿ�����ձ���������ð����ֹͣ���Ⱥ��������һ��ˮ���������������� ����ȫƿ ����β���е����� ����Ũ�� ��ȴ�ᾧ ������������ֽ� A BC
��������
ii.������ص��Ʊ�������ϡ����ʹ���Ʒ�Ӧ�������ᣬ����ʹ����ӷ�������װ�ã���ˮԡ���ȣ�����������ˮ��������װ�û���ȫƿ��ֹ���������ã��ڱ��н��������£������������ط���3K2MnO4+4CH3COOH=2KMnO4+MnO2��+4CH3COOK+2H2O����Ϊβ������װ�ã���������������ȣ�����ܿ��Է�ֹ������
(1)ʵ���м����ۻ��������õ�����Ϊ����������Ϊ��Ӧ���е�ǿ��KOH����������еĶ������跴Ӧ���Ӷ�����������Ӧѡ��������������ѡD��
(2)����ʵ��װ�õ������Լ�飬���õ�����������������ԭ����ʹװ���γ��ܱ���ϵ��������Ⱥ۲��Ƿ��������ݳ�����ȴ�������������Ƿ��γ�һ��ˮ�����ʼ����װ�������Եķ���Ϊ���Ӻ�װ�ã��رշ�Һ©�����������ձ��м���ˮ���þƾ�����������ƿ�����ձ���������ð����ֹͣ���Ⱥ��������һ��ˮ���������������ã�
(3)��������������ˮ������������������װ����ȫƿ��ֹ���������ã���Ӧ���������ӷ���ʵ��������ֶ�װ���ҽ�����ˮԡ���ȣ���һ���ٽ�������Ļӷ�������β���к������ᣬ����NaOH��Һ�����գ�
(4)�ȹ��˳�ȥ�������̣��ٶԸ��������Һ����Ũ����Ȼ����ȴ�ᾧ�����ɵõ�������ؾ��壻���ڸ���������ʲ��ȶ��������ֽ⣬���Ը�����ؾ���ֻ�ܵ��º�ɣ�
(5)���Ը��������Һ��ǿ�����ԣ�Ӧѡ����ʽ�ζ��ܣ�����ѡA��
(6)A���ζ�ʵ���У���ƿ��������ˮ��Ӱ������������ᷴӦ���Խ����Ӱ�죬A��������⣻
B��ʢ������Һ�ĵζ��ܵζ�ǰ���첿�������ݣ��ζ��յ�ʱ�����ݣ�ʹ�ö���ƫ�����IJ������ƫ��ʹ����KMnO4Ũ��ƫ�ߣ�B��������⣻
C���ζ�ʱ������Һ����ƿ�⣬���IJ������ƫ��ʹ����KMnO4Ũ��ƫ�ߣ�C��������⣻
D��ʢװKMnO4��Һ�ĵζ�����ˮϴ��δ��KMnO4��Һ��ϴ���൱�ڶ�KMnO4��Һϡ�ͣ���ȡһ�������Һ������KMnO4���٣����IJ������ƫС��ʹ����KMnO4Ũ��ƫ�ͣ�D��������⣻
��������ѡBC��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�����Ŀ��ij�о���ѧϰС���������̿�(��Ҫ�ɷ���MnO2)���չ�ҵ�����е�SO2�����Ʊ������̵�������������(����Һ��pH<2�����г���Mn2+�⣬����������Fe2+��Al3+��Ca2+�Ƚ�������)��
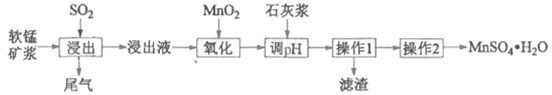
������1������Һͨ����������������ȥCa2+�����������γ������������ʱ��pH���±���
���� | ��ʼ����ʱ��pH | ��ȫ����ʱ��pH |
Fe2+ | 7.6 | 9.7 |
Fe3+ | 2.7 | 3.7 |
Al3+ | 3.8 | 4.7 |
Mn2+ | 8.3 | 9.8 |
(1)���������б����������ʵĻ�ѧʽΪ____________��
(2)������������Ҫ��Ӧ�����ӷ���ʽΪ_____________
(3)���������Һ���м���ʯ�ҽ�����pH�� pH �ķ�Χ��__________��
(4)����2��������Ũ����__________�� ���ˡ� ϴ�ӡ�����Ȳ�����
(5)Ϊ�ⶨ����MnSO4��H2O�Ĵ��ȣ�ȷ��ȡ������Ʒ1.720 g��������H2SO4��NH4NO3��Һ������ʹMn2+ȫ��������Mn3+�����200 mL��Һ��ȡ20.00 mL����Һ����������μ���0.0500 mol��L-1��FeSO4��Һ���������·�Ӧ�� Fe2+ +Mn3+=== Fe3+ + Mn2+����Ӧǡ����ȫ����ʱ��������FeSO4��Һ19.50 mL������MnSO4��H2O�Ĵ���(д��������̣����������λ��Ч����)__
����Ŀ��������ʵ����˵��Ԫ�صĽ����Ի�ǽ��������ǿ������
ѡ�� | ��ʵ | ���� |
A | ����ˮ��Ӧ,Na��Mg���� | ������:Na>Mg |
B | Ca(OH)2�ļ���ǿ��Mg(OH)2 | ������:Ca>Mg |
C | SO2��NaHCO3��Һ��Ӧ����CO2 | �ǽ�����:S>C |
D | t��ʱ,Br2+H2 I2+H2 | �ǽ�����:Br>I |
A. A B. B C. C D. D