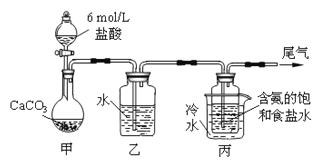
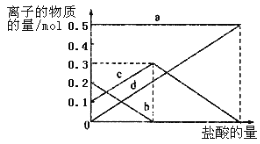
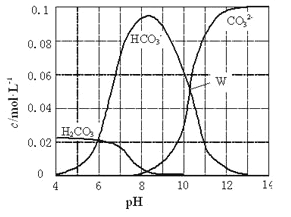
��Ŀ����
����Ŀ�������£���0.10mol��L-1��NaOH��Һ�ֱ�ζ������Ϊ50.00mL��Ũ�Ⱦ�Ϊ0.05mol��L-1��HClO��HF������Һ����ζ�������ͼ��ʾ��

����˵����ȷ����
A. HClO�����Ա�HFǿ
B. V(NaOH��Һ)=0 mL ʱ����Ӧ��Һ�У�c(ClO-)=c(F-)
C. V(NaOH��Һ)=12.50 mL ʱ����Ӧ��Һ�У�c(ClO-)>c(F-)
D. V(NaOH��Һ)=25.00mLʱ����Ӧ��Һ�У�c(ClO-)<c(F-)
���𰸡�D
��������A,��ͼ����Կ�����ͬ��0.10mol/L��һԪ�ᣬHClO��pHԼΪ2����HF��pHԼΪ4������HF���Ա�HClOǿ��A����ȷ��B��������������֪��HF���Խ�ǿ�������Ƚϴ�����������F-��Ũ��Ҳ�ϴ�B����C��V(NaOH��Һ)=12.50 mL ʱ������Һ��c(Na+)��ȣ�HF��Һ��pH<7�����ԣ���HClO��Һ��pH>7�Լ��ԣ��ɵ���غ��֪��c(Na+)+c(H+)=c(F-)+c(OH-), c(Na+)+c(H+)=c(ClO-)+c(OH-),����Һ��c(H+)Խ��c(OH-)��ԽС������c(ClO-)<c(F-)������C����D��V(NaOH��Һ)=25.00mLʱ����Ӧ��Һ�����ʷֱ�ΪNaClO��NaF����������ǿ���Σ��������ʵ���Ũ����ͬ������ˮ�����Һ���Լ��ԣ�����Խ��Խˮ���֪��ClO-��ˮ��̶Ƚϴ�����c(ClO-)<c(F-)��D��ȷ��